NOSB members were tentatively scheduled to make a decision Friday on whether to approve the continued use of “bioponics” to produce organically certified food. Bioponics combines the production methods of hydroponics and aquaponics to grow produce using containers but without using soil.
 Instead, the board voted 10-4 Friday afternoon to send the controversy back to a subcommittee for more discussion and clarification of how such production processes are defined. This means that food grown using hydroponic methods may continue to be certified as organic if the production operations otherwise meet established criteria for the designation.
Instead, the board voted 10-4 Friday afternoon to send the controversy back to a subcommittee for more discussion and clarification of how such production processes are defined. This means that food grown using hydroponic methods may continue to be certified as organic if the production operations otherwise meet established criteria for the designation.
The debate about whether carrageenan, an FDA-approved additive made from red seaweed, can safely be used in human food has been going on for years. Carrageenan is commonly used as a thickener and stabilizer in infant formula, dairy products, non-dairy “milk” analogs, meats and drink mixes.
Concerns have been raised about the risk of inflammation and possibly colon cancer in humans and animals from ingesting carrageenan in food products. However, there is a lack of consensus about whether the scientific studies done to date are conclusive about the effect on human health of consuming carrageenan.
Along with several other substances, it was scheduled to sunset from the National List of Allowed and Prohibited Substances in November 2018.
Reaction to the NOSB vote was swift, with industry groups expressing opposition and organic agriculture supporters declaring victory, although both sides noted that a final decision on the matter rests with USDA.
“The International Food Additives Council is disappointed the NOSB ignored overwhelming testimony that carrageenan is essential in the production of hundreds of organic foods and beverages,” said IFAC Executive Director Robert Rankin in a written statement.
Calling the NOSB vote “a concerning precedent,” Rankin added that delisting the substance will mean fewer options and inferior products as companies “struggle with alternatives that do not work as well as carrageenan.”
 Leon Bruner, chief science officer for the Grocery Manufacturers Association, said carrageenan should remain on the list of approved food additives because it has been proven safe for consumption and no alternatives provide the same functions.
Leon Bruner, chief science officer for the Grocery Manufacturers Association, said carrageenan should remain on the list of approved food additives because it has been proven safe for consumption and no alternatives provide the same functions.
“Regulatory agencies and research organizations around the world have consistently determined carrageenan is a safe and highly functional food additive,” Bruner said.
Susan Finn of United 4 Food Science said she was discouraged that the board “bowed to activist pressure and dubious science” in its vote to remove carrageenan from the list.
“The Board’s recommendation would make it difficult for organic food products to compete with non-organic products on sensory attributes such as taste and texture. This outcome may lead to consumers deselecting organic foods altogether, which runs counter to the National Organic Program’s mission,” she said.
Finn said the coalition group will now focus on convincing USDA to reverse the board’s decision.
FMC Corporation, a chemical manufacturer based in Philadelphia, released a statement that the company remains “fully supportive of carrageenan despite a deeply flawed decision” by the board.
“The NOSB’s decision serves as an interim recommendation and is not final,” FMC stated. “The final review and decision will be made by the United States Department of Agriculture, which has supported carrageenan’s use in food in the past. The USDA is scheduled to publish its final rule in November 2018.”
In celebrating the board’s decision, the Center for Food Safety said in a statement that consumers have been expressing their discontent with the presence of carrageenan in foods “so ardently” that some manufacturers have already abandoned the ingredient.
“The board’s recognition that carrageenan is not essential to organic, as evidenced by the ability of many manufacturers to eliminate it from their products, is an enormous victory for organic integrity,” said Cameron Harsh, CFS’ senior manager for organic and animal policy. “With this important vote, it is now in USDA’s hands to issue a proposed rule change for notice and comment as the next step in the removal process.”
 Mark Kastel, co-director of The Cornucopia Institute, said his organization was “very heartened that the NOSB voted to protect the reputation of the organic label.”
Mark Kastel, co-director of The Cornucopia Institute, said his organization was “very heartened that the NOSB voted to protect the reputation of the organic label.”
He added that the board’s action illustrates the power organic consumers have in the marketplace when they become active and vote with their wallets.
“We expect the USDA will respect the input of the organic community, and decision of the board, by publishing a rule in the Federal Register removing carrageenan and allowing a reasonable amount of time to clear existing inventories and reformulate products containing the ingredient,” Kastel said.
At this week’s meeting, the NOSB also rejected petitions to approve 1-Methylcyclopropene (1-MCP) for use on post-harvest apples to delay aging and ripening and the use of soy wax as plugs and sealant in organic mushroom production.
The NOSB is an citizen advisory group responsible for considering and making recommendations to USDA on issues involving the production, handling and processing of organic products. Among other duties, the board reviews each substance on the National List every five years to make sure it confirms to specific criteria and also consider petitions or add or delete a particular substance.
Members of the NOSB are appointed to five-year terms by the secretary of agriculture. It currently has four members who own or operate an organic farming operation; two who own or operate an organic handling operation; one who owns or operates a retail outlet trading in organic products; three with expertise in environmental protection; three from public or consumer interest groups; one with toxicology, ecology, or biochemistry expertise, and one who is a USDA-accredited certifying agent.
On Wednesday, Secretary of Agriculture Tom Vilsack appointed five new members to the NOSB who will begin serving their five-year terms on Jan. 24, 2017.
His appointees are: Asa Bradman, a California environmental health scientist; Steve Ela, who has an organic farm in Colorado; Sue Baird, an organic consultant in Missouri; David Mortensen, a plant ecology and weed management professor at Penn State, and Joelle Mosso, a product line manager for Olam International in Fresno, CA.
The new members will replace Carmela Beck, organic program manager for Driscoll Strawberry Associates in Watsonville, CA; Tracy Favre, a sustainable land management consultant from Texas; current NOSB Chair Jean Richardson, a retired University of Vermont professor of environmental studies and environmental law; Harold Austin of Zirkle Fruit Company, Selah, WA, and Zea Sonnabend, a California organic farmer and farm inspector.
(To sign up for a free subscription to Food Safety News, click here.)
]]>All patients are recovering from what was described as vomiting, diarrhea, stomach pain and some fever, according to the statement.
Health officials interviewed patients, obtained stool samples, inspected the food source and obtained food samples for testing at the state laboratory.
“The laboratory has reported that initial patient specimens tested presumptive positive for Salmonella. Food results are pending as it takes longer to process food specimens,” the statement noted.
The caterer, which has been reported as Indelible Catering of Moulton, AL, is cooperating with the health department and is no longer preparing food, according to the statement.
“The health department’s priorities are the health of the patients, a thorough investigation, and preventing any additional illness,” said Dr. Karen Landers, assistant state health officer. “This was a private event with exposure to the food over a specific time period. Therefore, there is no threat to the general public.”
Previous coverage follows:
Public health officials in northern Alabama are working to track down the source of a foodborne illness that has caused at least 40 people to seek medical attention and six to be hospitalized since Saturday night.
According to the Alabama Department of Public Health, officials started investigating the situation on Monday after reports emerged that a number of people had experienced bouts of diarrhea, vomiting, fever and abdominal pain following a weekend event in Sheffield, AL, near Muscle Shoals.
 “Currently we have about 40 people reported ill, although we expect that number to change. We have six hospitalized, and that may change, and probably will change, depending on how individual patients respond,” said Dr. Karen Landers, assistant state health officer.
“Currently we have about 40 people reported ill, although we expect that number to change. We have six hospitalized, and that may change, and probably will change, depending on how individual patients respond,” said Dr. Karen Landers, assistant state health officer.
Speculation was that contaminated food served at a catered affair in Colbert County may have been the source of the problem, although health officials were not sharing details about the venue, the caterer, or the food items involved.
“This was a private, catered event, and there were 150 people who attended,” Landers told Food Safety News.
Local news reports indicated the event was a hotel wedding reception in the Shoals region, and a story published Wednesday by The TimesDaily of Florence, AL, reported the source of the reception food as Indelible Catering.
Food samples and patient stool samples are currently being analyzed in the state laboratory, and preliminary results could be available by Thursday, Landers said.
“We have a menu (from the event), and it is appropriate to check the entire menu and what patients ingested. We should not focus on one food agent,” she said.
Landers would not speculate about whether the source of the problem is Salmonella, as some Alabama media outlets have reported.
“Many things can cause foodborne illness,” she said. “What I will say is once we have laboratory confirmation, we will provide more information. Our concern is for the patients, and that is being done, and we have mitigated or stopped any risk to the general public.”
The TimesDaily noted that the same catering firm provided food for a luncheon in 2014 in Decatur, AL, after which at least 19 people reported becoming ill. One of them died.
Several of those sickened were later confirmed as having E. coli and Salmonella infections. However, none of the food prepared for that 2014 luncheon by Indelible Catering was ever definitively linked to the reported illnesses.
(To sign up for a free subscription to Food Safety News, click here.)
]]>
The NOSB is an citizen advisory group responsible for considering and making recommendations to USDA on a wide range of issues involving the production, handling and processing of organic products. It has particular responsibility for adding, removing, or sunset reviewing items on the National List of Allowed and Prohibited Substances.
The list details a variety of substances that may and may not be used in organic production or handling. As part of its charge, the NOSB must review each substance on the list every five years to make sure it confirms to specific criteria and also consider petitions to add or delete a particular substance.
According to Miles McEvoy, deputy administrator of USDA’s National Organic Program, federal organic standards are designed to “allow natural substances in organic farming while prohibiting synthetic substances.” However, he notes there are exceptions, such as pheromones used on fruit crops to fight insects and certain animal vaccines.
“The National List also allows certain processing aids, such as baking soda. This substance lightens (or leavens) the dough for organic pancakes, baked goods, and other products,” McEvoy explains. “Conversely, some substances like strychnine and arsenic are examples of natural toxic substances that are prohibited in organic production.”
The carrageenan controversy
NOSB members will finish reviewing substances with 2018 sunset dates at this week’s meeting. One of them is carrageenan, an FDA-approved food additive made from a species of red seaweed and used in infant formula, dairy products, non-dairy “milk” analogs, meats and drink mixes to promote gel formation and thicken, stabilize and improve palatability and appearance.

Carrageenan is scheduled to be removed from the list on Nov. 3, 2018. It is currently categorized as a “nonagricultural substance” that may be used as an ingredient in or on processed products labeled as “organic” or “made with organic” ingredients or food groups.
Concerns have been raised about how the seaweed is grown and harvested, how it is processed, and whether carrageenan is a “synthetic material” under NOSB’s definition.
Concerns have also been raised about the risk of inflammation and possibly colon cancer in humans and animals from ingesting carrageenan in food products. However, there is a lack of consensus about whether the scientific studies done to date are conclusive about the effect on human health of consuming carrageenan.
According to a report released earlier this year by The Cornucopia Institute, all food-grade carrageenan is dangerous because it contains a carcinogenic contaminant — low molecular weight poligeenan.
However, Marinalg International, which represents the seaweed industry, counters that poligeenan is a “similar but wholly distinctive substance from carrageenan that is never used in foods.”
A subcommittee of NOSB has stated that it is aware of the ongoing scientific debate, that carrageenan has already been removed from many food products in the past few years, and that “suitable alternatives” are available such as gellan gum, xanthan gum and guar gum.
Because alternatives are available, the subcommittee voted 5-2 earlier this year to propose removal of carrageenan from the National List.
The debate over ‘bioponics’
Also on the NOSB agenda this week is the question of whether produce grown using “bioponics” should be eligible for organic certification. Bioponics combines the production methods of hydroponics and aquaponics to grow produce using containers but without using soil.
The debate is especially complex because crops raised using something other than soil have been eligible for organic certification since USDA established the National Organic Program in 2002.
Advocates claim that today’s bioponics methods use less water and fertilizer and enhance food safety and sustainability, while opponents say that bioponics excludes soil-plant ecology essential to organic farming methods and bypasses the age-old tradition of crops being grown in the ground.
A 16-member task force established last year by the National Organic Program reported back to the NOSB in July with an extensive report about current technologies and practices being used and examining whether and how they align with the Organic Foods Production Act and USDA organic regulations.

In essence, the task force supported “the historical premise that organic production should take place in soil,” but noted that the decision is ultimately up to the NOSB.
“This will be a very important recommendation for the future of organic certification,” the task force report stated. “No matter what one thinks about which path is best, we can all accept that many in the organic community are opposed to the inclusion of hydroponic as organic. Failure to address that concern will inevitably undermine public and farmer support for the USDA Organic label.”
Members of the NOSB are appointed to five-year terms by the secretary of agriculture. It currently has four members who own or operate an organic farming operation; two who own or operate an organic handling operation; one who owns or operates a retail outlet trading in organic products; three with expertise in environmental protection; three from public or consumer interest groups; one with toxicology, ecology, or biochemistry expertise, and one who is a USDA-accredited certifying agent.
The NOSB plans to meet Wednesday through Friday at the Chase Park Plaza Hotel in St. Louis. The full agenda can be found here.
(To sign up for a free subscription to Food Safety News, click here.)
]]>The samples were collected by U.S. Food and Drug Administration inspectors who visited the Simply Fresh Fruit Inc. plant in April and May, and Whole Genome Sequencing analysis determined the four isolates to be the same strain of Listeria.
According to a warning letter dated Oct. 19 and sent from FDA’s Los Angeles District Office to William T. Sander, company president, two of the positive environmental swabs were collected in areas adjacent to food contact surfaces.
 These included the panel surface and power switch on the side of a wash bin at the south end of a production line and a power knob for an upper conveyer belt at the north end of the same production line, FDA wrote.
These included the panel surface and power switch on the side of a wash bin at the south end of a production line and a power knob for an upper conveyer belt at the north end of the same production line, FDA wrote.
The third positive swab was collected from the blade of a squeegee used to push excess water into the drain on the floor, the warning letter stated.
FDA noted that finished product from the facility was also found to be positive for Listeria this past spring.
“Your firm filed a reportable food report (RFR) and conducted a voluntary recall of a cut cantaloupe lot processed on March 28, 2016, and packed on March 29, 2016, due to a L. monocytogenes positive finding,” according to the warning letter.
While the Simply Fresh Fruit website details some of the company’s food safety practices, HACCP plan and inspection protocols, there was nothing posted Tuesday under a “Product Alerts” tab.
There was also no FDA notice posted regarding the company’s April 14 recall of cantaloupe and fruit mixes with cantaloupe for possible Listeria contamination, nor was that news evident on the company’s website. However, a recall notice was posted at the time by US Foods and also by the California Department of Public Health.
Simply Fresh Fruit previously recalled fresh-cut fruit products with cantaloupe in March 2008 and cut fresh fruit trays in March 2007, although those recalls were for possible Salmonella contamination of products from outside suppliers.

In its responses to FDA, Simply Fresh Fruit blamed the current Listeria problem on the plant’s floor, according to the warning letter.
“You have determined your root cause to be ‘the condition of your dairy tile flooring’ and have implemented more aggressive sanitation procedures for your floors and have made corrections to your floors. We will ascertain the adequacy of your corrective actions during our next inspection,” the agency wrote.
FDA inspectors also observed a number of violations of Current Good Manufacturing Practice regulations at the Vernon processing plant, according to the warning letter.
Employees using gloves hands to cut melons then touched equipment and resumed handling fruit without cleaning and sanitizing the gloves, uncovered bins of washed fruit were stored in rooms during cleaning and sanitation operations, bin dumpers were in direct contact with wet floors, and forklifts were going through standing water to deliver pre-washed fruit to receiving areas, thereby potentially causing backsplash onto equipment and fruit products, FDA wrote.
While the company’s response to FDA addressed worker training on proper fruit storage, it did not address training regarding cross-contamination, the warning letter pointed out.
Further, the company failed to clean and sanitize food-contact surfaces in wet processing before use, and citrus build-up was observed on equipment during pre-washing and sanitation of cantaloupes, according to the agency.
“Your firm’s response states that fruit build-up will be removed upon each changeover of fruit and the drain wash bin will be drained and refilled with sanitizer solution. We will ascertain the adequacy of your corrective action during our next inspection,” the warning letter stated.
 FDA also noted that floor drainage was inadequate in areas where flooding-type cleaning was done or where normal operations release or discharge water or other liquid waste on the floor.
FDA also noted that floor drainage was inadequate in areas where flooding-type cleaning was done or where normal operations release or discharge water or other liquid waste on the floor.
Specifically, standing water was observed near the drain in the low-care area of the processing facility and near a scrub brush waster machine used to pre-wash and sanitize whole fruits, the agency wrote.
FDA gave the company 15 working days after receiving the warning letter to respond with specific steps it is taking to correct the violations, including an explanation of how the firm plans to prevent future violations, or similar ones, from recurring.
Listeria monocytogenes is a pathogenic bacterium that is widespread in the environment and may be introduced into a food processing facility from raw materials, humans, or equipment, according to FDA. Without proper controls, it can proliferate in food processing facilities where it may contaminate food, the warning letter noted.
Consuming these contaminated foods can lead to a severe and sometimes life-threatening illness called listeriosis, which is a major public health concern due to the severity of the disease, its high case-fatality rate, long incubation time and tendency to affect individuals with underlying conditions, FDA stated.
(To sign up for a free subscription to Food Safety News, click here.)
]]> While county health officials can’t be sure what caused the recent sicknesses linked to Fiesta Cancún, they levied a $250 fine, put the restaurant on probation for six months, and plan to do monthly inspections during that time.
While county health officials can’t be sure what caused the recent sicknesses linked to Fiesta Cancún, they levied a $250 fine, put the restaurant on probation for six months, and plan to do monthly inspections during that time.
Food samples taken from the restaurant and tested by the Indiana Department of Health were all negative for pathogens, according to Carolyn Smith, food service director for the St. Joseph County Health Department in South Bend.
“All the samples came back, and nothing was found,” Smith said. She added that the state health department realized the samples brought in for testing weren’t food items actually consumed by those who reported becoming ill after eating at Fiesta Cancún on Oct. 15, 19 and 21. The restaurant had only been open since Oct. 3.
“This was about a week later, but if there were practices, then the thinking from the Indiana State Department of Health was that we still might find something due to those practices,” Smith said.
She explained that the Indiana food code allows health officials to temporarily close a restaurant if there is an apparent foodborne illness outbreak involving two or more people who have ingested common food items.
A number of calls had come in to the county health department complaining about illnesses possibly linked to the restaurant not long after it opened. First there was just one party and then two, Smith recalled.
“Then a lady called and said there were seven adults in her party (who were sickened),” she said. However, she added, their healthcare providers didn’t test any of them.
One man called Oct. 16 to complain about vomiting, diarrhea and stomach cramps that he reported experiencing after dining the previous evening at Fiesta Cancún. Following his complaint, an inspection on Oct. 18 identified eight health code violations, five of them rated critical.
Fiesta Cancún was closed Oct. 25. A hearing on the situation was held Oct. 31, the same day another inspection was done, Smith said.
 Meanwhile, the restaurant had disposed of all food items during the course of the six days the doors were closed and had already adjusted some of its practices, she said.
Meanwhile, the restaurant had disposed of all food items during the course of the six days the doors were closed and had already adjusted some of its practices, she said.
“They had made major changes in terms of the flow of food in their operation and created a room dedicated specifically for prepping. They had a meeting with their staff and also meetings with their staff and one of our staff members who went in and did some training,” Smith said.
A Facebook message posted Oct. 31 by the restaurant thanked customers for their patience, trust and support and noted that the doors would reopen at 2 p.m. on Nov. 1.
“If you previously had a bad experience at Fiesta Cancún give us the chance to gain your business back by letting us make it right/better for you,” the message read.
Smith noted that the restaurant’s owners did more than was required to get back into compliance with county and state food regulations.
“This group of gentlemen who run this restaurant were over and above on what they need to comply and the changes that they’ve made,” she said, indicating that the business had quickly become popular and inundated with customers.
A number of issues that cropped up during the outbreak and subsequent investigation made it hard for food safety officials to know exactly what occurred and whether specific food items served at Fiesta Cancún caused the reported illnesses.
The cases weren’t confirmed by laboratory testing, the food items weren’t collected and tested until several days after reports of illness, and nothing found in the restaurant could specifically be tied to foodborne pathogens.
The resulting frustration was evident when Nick Molchan, administrator of the St. Joseph County Health Department, told the South Bend Tribune that the county would like to send this message to local doctors:
“Hey, when you have somebody in and you think it might be foodborne illness, take a stool sample!”
 The study, from researchers at the Centers for Disease Control and Prevention, was published online Nov. 1 in the Journal of Food Protection.
The study, from researchers at the Centers for Disease Control and Prevention, was published online Nov. 1 in the Journal of Food Protection.
Using CDC’s Foodborne Disease Outbreak Surveillance System, researchers identified 18 outbreaks between 1992 and 2014 that were reportedly caused by organic food products. These outbreaks were linked to 779 illnesses, 258 hospitalizations and three deaths.
More than half of the total outbreaks, 56 percent, occurred from 2010-2014, which the study authors note reflects the increasing production and consumption of organically grown food in the United States and around the world. Nine of the outbreaks occurred in a single state, while nine were multistate.
The most commonly occurring pathogens were Salmonella, responsible for 44 percent of the outbreaks, and E. coli O157:H7, the pathogen in 33 percent of the outbreaks, according to the study. Campylobacter, Clostridium botulinum and the Hepatitis A virus caused one outbreak each.
Eight of the 18 outbreaks were attributed to produce items, four to unpasteurized dairy products, two to eggs, two to nut and seed products, and two to multi-ingredient foods. Fifteen of the 18 outbreaks were associated with foods that were definitely or likely U.S. Department of Agriculture-certified as organic.
“Consumer perception often credits organic foods as being safer than conventionally produced foods, although organic standards do not directly address safety issues such as microbial or chemical hazards,” according to the study abstract.
 The term “organic” had no official definition until 2000 when USDA’s National Organic Program standards were published, said Sam Crowe, epidemiologist and team lead for CDC’s National Outbreak Reporting System.
The term “organic” had no official definition until 2000 when USDA’s National Organic Program standards were published, said Sam Crowe, epidemiologist and team lead for CDC’s National Outbreak Reporting System.
“So foods described as ‘organic’ before that time may not mean the same thing as it does now,” he told Food Safety News.
Crowe said information about how food is produced, whether conventional or organic, is not systematically collected through the National Outbreak Reporting System (NORS) and therefore it’s difficult to compare outbreaks linked to differing production methods.
“The authors had to search each outbreak report from 1973, when CDC began collecting information on foodborne outbreaks, through 2014 for any mention of the word ‘organic’ in order to identify organic food outbreaks,” he said.
“Some additional outbreaks could have been from food that was organically grown but not listed as such in the outbreak report. So we cannot say for sure how many outbreaks were from conventionally grown foods.”
Because of this current lack of regular data collection, the CDC researchers couldn’t pin down the relative risk of foodborne illness.
“We are unable to assess risk of outbreaks due to organic foods compared with conventional foods because foodborne outbreak surveillance does not systematically collect food production method,” the study states.
 Crowe acknowledged there may be benefits to collecting information on how foods associated with outbreaks were grown. However, he added that local and state health department outbreak investigators are often responsible for collecting it during outbreak investigations, and that it is frequently not readily available.
Crowe acknowledged there may be benefits to collecting information on how foods associated with outbreaks were grown. However, he added that local and state health department outbreak investigators are often responsible for collecting it during outbreak investigations, and that it is frequently not readily available.
“These investigators already have a tremendous amount of responsibility and local and state health departments are often understaffed and underfunded, so the additional burden would need to be assessed to determine whether or not this information is worth adding to the long list of requirements,” he said.
“At this time, CDC does not have plans to change the outbreak reporting system to capture this information, but nevertheless will continue to explore ways to improve outbreak reporting.”
Despite the paucity of consistent and relevant data, the researchers did offer some recommendations for consumers who want to avoid being sickened by foodborne pathogens, whether their foods of choice are organic or conventional.
“Consumers should be aware of the risk of milk and produce consumed raw, including organic,” they wrote. “Consumers should not assume organic foods to be more or less safe than foods produced by conventional methods. Proper handling, preparation, and storage of foods, regardless of production method, are necessary to prevent foodborne illness.”
The authors were R. Reid Harvey, Christine M. Zakhour and L. Hannah Gould, who were all with CDC’s Division of Foodborne, Waterborne, and Environmental Diseases at the time of the study.
(To sign up for a free subscription to Food Safety News, click here.)
]]>
CDC investigators are now on the scene in Flint, MI, to help track what caused the outbreak of this highly contagious gastrointestinal disease and see what can be done to prevent further illnesses.
They are busy reviewing the known cases and checking into potential connections with those being reported from other parts of Michigan, according to a community update posted Oct. 26 from CDC, state and county health officials.
“We’re contacting people who were ill in Genesee and Saginaw County to figure out how this was spread. There are so many possible cases we’re not speculating,” said Genesee County Health Division Director Suzanne Cupal.
The number of shigellosis cases in the area is much higher this year than in previous years, she said, peaking in early July and slowing down since September. At least 27 of those sickened have been hospitalized, but no related deaths have so far been reported.
There has been speculation that the shigellosis outbreak could be linked to the much-publicized water problems in Flint. That’s because some residents are afraid to use the tainted city water to wash their hands and therefore could be allowing foodborne and waterborne pathogens to gain a foothold.
Cupal and her public health colleagues are advocating thorough and frequent handwashing to help prevent further spread of the disease.
“Some germs like Shigella only take a small amount to make you ill,” she said. “This is an opportunity to remind everyone that hand-washing should be a healthy habit you practice every day. It’s critical after you’ve diapered, used the bathroom and before you cook food.”
If hand sanitizer is used instead, it should contain at least 60-percent alcohol, Cupal advised.
 “We want everyone to make handwashing a healthy habit that everybody does regularly,” she said. “Use soap, and rub hands together for at least 20 seconds. The friction with the foam is what’s getting the bacteria off your hands.”
“We want everyone to make handwashing a healthy habit that everybody does regularly,” she said. “Use soap, and rub hands together for at least 20 seconds. The friction with the foam is what’s getting the bacteria off your hands.”
Shigellosis is a highly contagious disease caused by four different strains of Shigella bacteria. Even a microscopic amount of contaminated fecal matter in food, water or other beverages can cause infection if consumed.
Most people infected with the bacteria develop diarrhea, fever and stomach cramps within a day or two after being exposed. The symptoms usually resolve within five to seven days.
Some people who are infected may have no symptoms at all, but may still pass the Shigella bacteria to others. The spread of Shigella can be stopped by frequent and careful handwashing with soap and by taking other hygiene measures.
If you or someone in your family has symptoms of Shigella infection such as diarrhea, a stomach ache and occasional fever, the Genesee County Health Department recommends visiting a healthcare provider and asking about a test for Shigella.
(To sign up for a free subscription to Food Safety News, click here.)
]]> The International Company for Agricultural Production & Processing (ICAPP), based in Ramadan City, Egypt, is voluntarily recalling frozen strawberries in response to the ongoing investigation into a multistate outbreak of Hepatitis A in the United States.
The International Company for Agricultural Production & Processing (ICAPP), based in Ramadan City, Egypt, is voluntarily recalling frozen strawberries in response to the ongoing investigation into a multistate outbreak of Hepatitis A in the United States.
The Food and Drug Administration’s recall announcement, dated Sunday, stated that the company’s action was taken in consultation with FDA because the Hepatitis A virus was detected in four lots of frozen strawberries ICAPP exported to the U.S.
The federal agency added that ICAPP is working closely with all of its distributors in this country to make sure that the recall is effective.
FDA issued an Import Alert for the company’s strawberries on Oct. 19, indicating that the frozen berries would not be admitted into the U.S. However, at the time the agency did not specifically connect the product to the Hepatitis A outbreak that has been linked to smoothies served at Tropical Smoothie Café outlets.
However, FDA’s latest update on the investigation, posted Oct. 20, notes the following: “Nearly all ill people interviewed report eating smoothies containing strawberries at Tropical Smoothie locations in a limited geographic area. Preliminary traceback information indicates that the frozen strawberries served in these Tropical Smoothie Café locations were imported from Egypt. Tropical Smoothie Café has stopped using these strawberries nationwide.”
 As of Oct. 17, the Hepatitis A outbreak had sickened 134 people from nine states — Arkansas, California, Maryland, North Carolina, New York, Oregon, Virginia, Wisconsin and West Virginia — according to the U.S. Centers for Disease Control and Prevention. No deaths have been reported in connection with the outbreak, although 52 people have been hospitalized.
As of Oct. 17, the Hepatitis A outbreak had sickened 134 people from nine states — Arkansas, California, Maryland, North Carolina, New York, Oregon, Virginia, Wisconsin and West Virginia — according to the U.S. Centers for Disease Control and Prevention. No deaths have been reported in connection with the outbreak, although 52 people have been hospitalized.
FDA noted in the Oct. 30 recall announcement that the lots of frozen Egyptian strawberries were all distributed for sale to, and use in, foodservice establishments nationwide and not for use in food products offered for retail sale to consumers.
Even so, the agency added that ICAPP was “issuing this news release publicly to help mitigate any possible risk to the public health and to fully ensure that all recalled products are recovered.”
“Although none of ICAPP’s own testing through an established surveillance program or through third party testing of retained samples has identified the presence of Hepatitis A in any of its products, ICAPP has decided to recall all frozen strawberries that it has imported into the United States since January 1, 2016 out of an abundance of caution,” according to the recall announcement.
No other ICAPP products, frozen or fresh, are covered by this voluntary recall, FDA added.
The recall announcement stated that the Egyptian company is conducting a comprehensive review of all of its operations and suppliers to make sure that the food it produces is safe.
For questions or more information about the recall, consumers may contact ICAPP by email at [email protected], or by phone, between 9 a.m. and 5 p.m. Cairo local time, which is six hours ahead of EDT, at +201-541-1624.
 Under the Food Safety Modernization Act (FSMA), FDA has mandatory recall authority, but the agency must first provide the responsible party with a chance to stop distribution and conduct a voluntary recall of the food item in question.
Under the Food Safety Modernization Act (FSMA), FDA has mandatory recall authority, but the agency must first provide the responsible party with a chance to stop distribution and conduct a voluntary recall of the food item in question.
If the responsible party refuses to, or does not voluntarily, cease distribution or recall the food item within the time and in the manner prescribed by FDA, the agency may proceed with a mandatory recall.
Hepatitis A is a contagious liver disease that results from exposure to the Hepatitis A virus. It is usually transmitted by the fecal-oral route, either through person-to-person contact or through consumption of contaminated food or water.
It can range from a mild illness lasting a few weeks to a serious illness lasting several months. Illness generally occurs within 15 to 50 days of exposure and includes fatigue, abdominal pain, jaundice, abnormal liver tests, dark urine and pale stool.
Hepatitis A vaccination can prevent illness if given within two weeks of exposure to a contaminated food. In rare cases, particularly for those who have a pre-existing severe illness or are immune-compromised, Hepatitis A infection can progress to liver failure.
Persons who may have consumed the recalled product should consult with their health care professional or local health department to determine if a vaccination is appropriate, and anyone with symptoms of Hepatitis A should contact their health care provider or a local health department immediately.
(To sign up for a free subscription to Food Safety News, click here.)

Fast casual restaurants are defined as those that do not offer full table service but promise higher-quality food with fewer frozen or processed ingredients than fast food restaurants. Some national chains in this category are Chipotle, Panera, Qdoba and Panda Express.
San Diego-based TrendSource Inc. conducted the survey between Aug. 11 and Sept. 25 by asking 2,969 people aged 18 and older about their perceptions of food safety, food cleanliness, food handling, employee cleanliness and other related issues during visits to fast-casual restaurants.
Most of the respondents were Caucasian, female, between 31-60 years old, had a college education and incomes between $25,001 and $100,000.
Some key findings:
- 65 percent said they eat at a fast casual restaurant a “few times a month” and visit about four different ones during that period;
- 81 percent said that seeing their food prepared increases their trust in its safety and quality;
- 86 percent said they feel their food is “safe” or “very safe” if it was prepared by people wearing gloves;
- 53 percent said they don’t believe restaurant employees wash their hands after using the bathroom or taking a smoke break and before handling food;
- 82 percent said they are “likely” or “very likely” to complain to a manager if they witnessed unsanitary employee behavior or conditions at a restaurant;
- 46 percent said they associated certain types of cuisine with foodborne illness. This “cuisine bias” was mainly directed at, in this order, Mexican/Latin, Asian, India, American and Mediterranean food;
- 70 percent don’t believe there is a lower risk of illness from food that is organic or free of GMOs and/or antibiotics;
- 56 percent said they are “unlikely” or “very unlikely” to return to a restaurant if someone they know became ill from eating there;
- 61 percent said they would return to a restaurant that had faced a foodborne illness outbreak, but most would wait a month or two and until friends ate there without incident and a health inspector or agency had declared the problems fixed; and
- 42 percent said they would complain on social media about food safety concerns, with 88 percent of those using Facebook, 37 percent using Yelp and 24 percent using Twitter to do so.
TrendSource Marketing Manager Heather Parker said that while the recent foodborne illness outbreaks linked to Chipotle Mexican Grill helped prompt the survey, the fast casual chain is not a client.
“We (the marketing team) do market research, so at any given time we’re looking at what people are talking about,” she explained.
TrendSource uses a closed database of about 300,000 “field agents” who may choose to respond to its electronic surveys. The topics change regularly, depending on what the general public happens to be discussing regarding a given industry.
Parker noted that the definition of a fast-casual restaurant varies, but the way TrendSource views the term is “any restaurant that allows you to order at a counter and does not have a drive-thru option.”
However, even that distinction is becoming blurred as more fast-casual chains, particularly regional ones, start installing drive-thrus.
For Parker, the “most fascinating part” of the entire survey was the 7o percent of respondents who said they don’t believe organic food or food free of GMOs and/or antibiotics poses a lower risk of illness.
“I would think that it would be healthier or would be safer, but for this study, that was not the case,” she said.
“The other thing, which wasn’t surprising but interesting, was that 81 percent said seeing their food prepared increased safety and quality,” Parker said, adding that consumers don’t typically see where the food comes from.
Fast casual restaurants are one of the fastest growing segment of the U.S. restaurant industry and are organizing their own events and conferences, such as the annual Fast Casual Executive Summit.
Some reports put fast casual sector growth in the U.S. at nearly 600 percent between 2000-2014, with the yearly growth rate almost double that of the restaurant industry as a whole.
(To sign up for a free subscription to Food Safety News, click here.)
]]>State food protection officials also notified businesses to recall any affected shellfish harvested in the area on or after Sept. 26, according to a public health advisory issued Oct. 13.
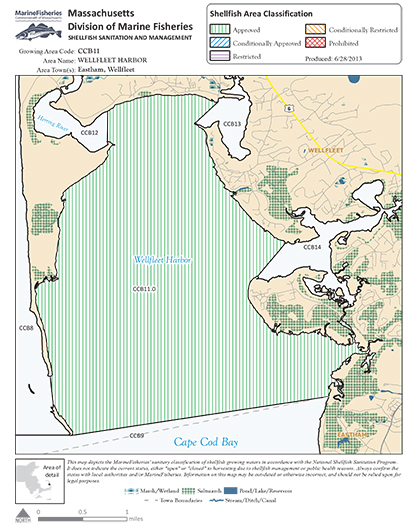
“Public health officials have received reports of approximately 75 suspect cases of norovirus over the past two days, primarily associated with eating raw shellfish at weddings and restaurants in the Outer Cape Cod area,” the advisory noted.
Harvesting of oysters and other shellfish in Wellfleet will be closed for at least three weeks, although other Cape Cod shellfish-harvesting areas are not affected. The currently closed areas include Wellfleet Harbor, Wellfleet Inner Harbor, Herring River and Loagy Bay.
“The National Shellfish Sanitation Program requires a minimum closure of 21 days,” said Katie Gronendyke, a spokeswoman for the Massachusetts Division of Marine Fisheries in Boston. “The state has to demonstrate that there are no ongoing pollution problems by conducting a resurvey of the area and an evaluation of all real and potential pollution sources.”
It’s not clear how many businesses might be involved in the recall because locally harvested shellfish — oysters as well as mussels and clams — are shipped all over the world, said Hillary Greenberg-Lemos, a health and conservation agent for the Town of Wellfleet.
The lack of raw oysters affected the 16th annual Wellfleet OysterFest held this past weekend. No raw shellfish were served at the event, and the popular “Shuck Off” contest used oysters sourced from outside the area.
“Public safety has always been our top priority, whether it be in crowd management or health issues,” said Alex Hay, a board member of the group that puts on the OysterFest, adding, “Not only is public safety our main concern, it is also in the seafood industry’s best interest, the town’s best interest and the Wellfleet brand’s best interest.”
Norovirus is easily spread through food, person-to-person contact, or through contact with contaminated surfaces such as countertops or door knobs. The viruses can cause vomiting, diarrhea, and abdominal cramps. Other symptoms may include headache, fever, chills and muscle aches.
For most people, noroviruses cause a very unpleasant but brief illness that usually lasts one or two days. The elderly, very young and people with weakened immune systems are at higher risk of more serious illness from noroviruses. The greatest danger is from dehydration.
 The Massachusetts Department of Public Health offered this safety advice to the public:
The Massachusetts Department of Public Health offered this safety advice to the public:
- Do not eat any shellfish from the affected areas if harvested or purchased on or after Sept. 26. This includes oysters, hard-shell clams (“quahogs”), soft-shell clams (“steamers”), mussels and razor clams.
- If you are unsure where shellfish was harvested, contact the retailer or restaurant where the product was purchased.
- If you are experiencing symptoms of norovirus, or are caring for someone who is, practice good hand washing with warm, soapy water to prevent further spread of the illness.
- After episodes of vomiting or diarrhea, clean contaminated surfaces immediately with a bleach-based household cleaner and wash hands thoroughly afterward. The virus is present in large numbers in both stool and vomit.
- Contact your healthcare provider if you have more severe illness or if you think you may be dehydrated.
For more information about noroviruses, Bay State residents can contact the Massachusetts Department of Public Health’s Division of Epidemiology at 617-983-6800. For more information on the shellfish-harvesting closure notice, call 508-990-2860.
According to a study published in August 2015 in the journal Applied and Environmental Microbiology, oysters act as incubators for noroviruses to mutate between outbreaks, and raw oysters appear to be particularly dangerous in this way.
“Shellfish such as oysters, clams, and mussels are filter feeders and ingest norovirus if it is present in the water. Through filter feeding, oysters may concentrate the virus to much higher levels than might be found in the surrounding water,” the Washington State Department of Health has reported.
“Potential sources of contamination include faulty wastewater treatment plants, failing septic systems, stormwater runoff, dumping of boat sewage waste, and vomiting overboard near shellfish beds.”
(To sign up for a free subscription to Food Safety News, click here.)
]]>
The FDA warning letter, dated Oct. 7, was addressed to Stewart Resnick, owner of The Wonderful Company based in Los Angeles, and concerned Wonderful Pistachios processed by Paramount Farms at 13646 Highway 33 in Lost Hills, CA, about 45 miles northwest of Bakersfield.
The outbreak sickened at least 11 people in nine states and hospitalized two of them. No deaths were reported, according to a final update from the U.S. Centers for Disease Control and Prevention posted May 20.
Among people for whom information was available, illnesses started on dates ranging from Jan. 3 to March 25. Ill people ranged in age from 27 years to 69, with a median age of 41. Sixty-four percent of those sickened were male, according to CDC.
On March 9, The Wonderful Company recalled various flavors and sizes of in-shell and shelled pistachios sold under the brand names Wonderful, Paramount Farms and Trader Joe’s. The recalled nuts were sold nationwide and in Canada, Mexico and Peru. The full list of recalled products is here.
Although CDC declared the outbreak investigation over back in May, since the recalled pistachios have a long shelf life and may still be in people’s homes, the agency noted that consumers unaware of the recall could continue to eat the products and get sick.
FDA’s San Francisco District Office in Alameda told Resnick in the warning letter that, following the Salmonella outbreak, investigators had inspected the company’s pistachio manufacturing process at the Lost Hills facility from March 8 through April 7.
“Based on traceback and epidemiological evidence taken together with inspectional evidence, we have concluded that pistachio nuts produced by your firm are adulterated within the meaning of … the Federal Food, Drug, and Cosmetic Act … in that they bear or contain Salmonella, a deleterious substance which may render them injurious to health …,” the letter stated.
“Based on collaborative epidemiological and investigational efforts between the FDA, CDC, and the California Department of Public Health, we conclude that pistachio nuts produced by Wonderful Pistachios & Almonds LLC were linked to this outbreak of Salmonella infections.”
Of the 11 people confirmed in the Salmonella outbreak, FDA stated that 10 were interviewed. Eight of the 10 reported eating pistachios during the week before they became ill, and five of those reported eating Wonderful brand pistachios. No other specific food was identified as a “suspect vehicle,” according to the warning letter.
Nuts from facility tested positive for outbreak strain
During FDA’s visit to Paramount Farms, inspectors collected three product samples, each consisting of 30 subsamples. In one sample of raw, in-shell pistachios collected from the facility’s silos, five of 30 subsamples yielded positive tests, four for Salmonella Senftenberg and one for Salmonella Liverpool, according to the warning letter.
 “Whole genome sequencing determined that the Salmonella Senftenberg isolates were nearly identical to isolates from case patients involved in the outbreak. The most probable number in these subsamples ranged from less than 3 to 23 Salmonella cells/gram,” FDA wrote.
“Whole genome sequencing determined that the Salmonella Senftenberg isolates were nearly identical to isolates from case patients involved in the outbreak. The most probable number in these subsamples ranged from less than 3 to 23 Salmonella cells/gram,” FDA wrote.
The agency’s letter acknowledged The Wonderful Company recall of pistachio products and its written response to the inspection observations dated April 19. In that response, the company proposed to study the optimal chlorine level in its processing tanks and install a system to remove foreign material prior to moving the pistachios into storage, the warning letter stated.
“However, you did not provide us with documentation demonstrating the effectiveness of these changes and any other changes you have made to prevent a reoccurrence of an outbreak,” FDA noted, adding that it would need documentation, including photographs, of corrective actions taken to date and any other pertinent information that might help evaluate such corrections.
As of Thursday, FDA had not yet heard back from Resnick, The Wonderful Company or Paramount Farms, said Sergio Chavez, a compliance officer with the agency’s Alameda office. However, he pointed out that they have 15 working days from receiving the letter to do so.
In response to a request from Food Safety News, Steven Clark, vice president of corporate communications for The Wonderful Company, provided this statement regarding the warning letter:
“Yes, we were surprised to receive this communication from the FDA since we have not heard from them since early April when they inspected our facility. Other than the Oct. 7 letter, neither the FDA nor the CDC advised us of any further action required on our part. Moreover, the CDC closed their investigation last May.
“Though none of the Salmonella cases in question were directly linked to our product, we did voluntarily recall product in an abundance of caution, we implemented enhanced food safety protocols, and no new issues have surfaced. We will continue to cooperate with the FDA to satisfy any concerns they may have.”
Chavez said that FDA can shut down a food facility by revoking its facility registration if it is found to present an imminent hazard to the public, adding, “That finding was not made in this particular instance.”
At this point, it’s not clear to FDA how the Salmonella bacteria got to the pistachios, but there are many potential ways it can happen, he said.

“If we knew how it got in there, we would have put it in that letter,” he noted. “Even if they (the nuts) don’t hit the ground, they’re out in nature and birds do what they do, lizards do what they do. Anything that’s on the nuts is a potential source.”
Chlorine will kill most pathogens, including Salmonella, as long as it is applied in the appropriate concentration and the level is monitored, the water regularly changed, and the chlorine gets directly to where the bacteria are hiding, Chavez explained.
“When you’re talking about cleaning and sanitizing something, germs will hide under anything, including dirt, and if you apply surface sanitizer and it doesn’t penetrate that gunk layer, no matter how thick it may be, it won’t fix the problem,” he said.
FDA will be reinspecting the Paramount Farms facility at some point, although Chavez could not say when.
“For food, we rarely tell people,” he said. “There’s supposed to be compliance all of the time, not just when we come by.”
In addition to Paramount Farms, Resnick and his wife Lynda also own POM Wonderful pomegranate juice, Halos brand mandarin oranges, Sweet Scarletts Texas red grapefruit, Fiji Water, the Teleflora flower-delivery service, Landmark and Justin wines, Suterra pest control, a large beekeeping farm and thousands of acres of California citrus, almond and pistachio orchards.
(To sign up for a free subscription to Food Safety News, click here.)
]]>Glyphosate is a widely used and controversial herbicide and the active ingredient in Monsanto’s Roundup. It was first licensed for use in the U.S. in the mid-1970s.
The test results were presented by Narong Chamkasem, a research chemist with FDA’s Southeast Regional Laboratory, at a scientific workshop this past July in Florida.

Chamkasem reported finding no glyphosate residues in organic oat samples from Bob’s Red Mill, Whole Foods, Sprouts and Nature’s Path, while other results from 10 unnamed brands ranged from 0.01 ppm in “apple cinnamon instant oat meal” to 1.67 ppm in “cinnamon spice instant oat meal.”
In 19 honey samples tested, he reported finding just a trace of glyphosate in 10 of them. Results from the other nine samples tested ranged from a low of 17 ng/g in Brazilian honey up to a high of 121 ng/g in honey from Louisiana.
The story was first reported by Carey Gillam here.
Maximum tolerance levels
The levels detected by the FDA testing are below the U.S. Environmental Protection Agency’s maximum tolerance level for glyphosate in oats, which is 30 ppm.
Other countries have lower maximum allowable limits on glyphosate residues in food and feed. The European Union, for example, has set a 20 ppm maximum residue level for glyphosate in oats.
There is currently no maximum tolerance level in the U.S. for glyphosate residues in honey, but the issue could land on the joint EPA/FDA agenda in the near future.
FDA started testing certain foods for glyphosate residues in February after the U.S. Government Accountability Office and others criticized the agency for not doing so and for not publicly revealing limitations in both its testing practices and its methodology.
Questions about safety
Because of potential links with certain types of cancer and reproductive and developmental problems, there are ongoing efforts to ban or limit the use of glyphosate in this country.
In March 2015, the World Health Organization’s International Agency for Research on Cancer classified glyphosate as “probably carcinogenic to humans” based on “convincing evidence that these agents cause cancer in laboratory animals.”
The Federal Insecticide, Fungicide, and Rodenticide Act Scientific Advisory Panel is meeting next week in Arlington, VA, to discuss the carcinogenic potential of the herbicide, and EPA is planning to issue related human health and ecological risk assessments next spring.
Meanwhile, in a “Glyphosate Issue Paper” released Sept. 12, EPA has proposed classifying glyphosate as “not likely to be carcinogenic to humans at doses relevant for human health risk assessment.” Comments on the proposal are being accepted until Oct. 17 here.
Monsanto’s position is that detectable glyphosate levels in human food are too low to cause any health problems and that testing ensures its safety.
“When it comes to safety assessments, no other pesticide has been more extensively tested than glyphosate. In evaluations spanning four decades, the overwhelming conclusion of experts worldwide, including the EPA, has been that glyphosate can be used safely according to label instructions,” the company states on its website.
In the past 20 years, the Missouri-based agrochemical giant has developed soybeans, sugar beats, canola and corn varieties genetically engineered as “Roundup Ready” to help make them immune to its branded glyphosate-based herbicide. The idea is to kill the weeds but not the crop.

The recent FDA testing results, the EPA risk assessment process, and the ongoing debate about the safety of glyphosate use on food and feed comes at a particularly delicate time for Monsanto.
The company was just purchased last month for $66 billion by German drug and agrochemical company Bayer, and the EU license for glyphosate was set to expire this year.
However, after some EU member states objected to renewing it, an eleventh-hour decision extended the license for 18 months — until the end of 2017 — pending further scientific study.
Quaker Oats lawsuit
A San Francisco woman filed a potential class-action lawsuit earlier this year against the Quaker Oats Co. alleging that the firm deceptively labels its products sold at retail as “100% Natural” when glyphosate is sprayed on the oats as a drying agent shortly before harvest.
The main plaintiff, Danielle Cooper, asked that the court order the company to issue refunds to purchasers of its products and to embark on a “corrective advertising campaign to inform the public of the true nature of Quaker’s carcinogen-contaminated oats.”
Quaker, which was bought by PepsiCo in 2001, stated that it does not add glyphosate during any part of the oat milling process, although the chemical is “commonly used by farmers across the country who apply it pre-harvest.” The company also noted that it “thoroughly cleanses” all oats it receives for processing.
“Any levels of glyphosate that may remain are trace amounts and significantly below any limits which have been set by the Environmental Protection Agency (EPA) as safe for human consumption,” Quaker stated on its website.
“It’s important to put this into perspective,” the Chicago-based company added. “The typical consumer would, on average, have to consume approximately 1,000 bowls of oatmeal a day to even come close the safe limit set by the U.S. government. We proudly stand by the safety and quality of all of our products. Producing healthy, wholesome food is Quaker’s number one priority and we’ve been doing that for nearly 140 years.”
(To sign up for a free subscription to Food Safety News, click here.)
]]>LGMA reported there were 470 announced and unannounced audits during last year and this year of its approximately 100 members, known as handlers, and that 385 citations of various types were handed out.
These audits are mandatory for LGMA members, who are audited an average of five times each year, with 185 checkpoints per audit.
On average, there were about 0.8 citations issued per audit. By category and percentage, they were given out for:
- Field Observations, 34 percent;
- Water Use, 26 percent;
- Worker Practices, 15 percent;
- Field Sanitation, 12 percent;
- Soil Amendments, 5 percent;
- Environmental Assessments, 4 percent; and
- General Requirements, 4 percent.
“For the past two years the average has been less than 1 citation per audit of an LGMA member,” according to Ron Ratto, president of Ratto Bros. Fresh Produce Inc. of Modesto, CA, and chairman of the LGMA advisory board.

Because the group already adopted and implemented food safety practices similar to federal requirements under the Food Safety Modernization Act’s (FSMA) Produce Safety Rule, Ratto added that “the leafy greens industry has had a nine-year head start on the new laws.”
A LGMA audit is based on seven categories of the group’s accepted food safety practices, which are called the LGMA Metrics.
For example, under the General Requirements category, handlers must meet specific requirements related to food safety management, including having a written compliance plan, a current list of growers, a documented traceability program, and two designated people available at any time to oversee the food safety program.
Ratto noted in the annual report that LGMA had a third-party review of the metrics done in 2015 by four nationally known food safety scientists. These reviewers found the metrics to be scientifically valid and to incorporate most current best practices, he stated.
“They also confirmed that the LGMA Metrics appear to meet or exceed the requirements of the new federal Produce Rule,” he said in the report.
The audit process Field inspections and audits of LGMA handlers in the Golden State are done by six or seven people from the California Department of Food and Agriculture (CDFA) who are licensed by the U.S. Department of Agriculture.
Steve Patton, chief of CFDA’s Inspection and Compliance Branch, said these auditors use the LGMA Metrics as a checklist when they visit leafy greens operations throughout the state.
“We go through and look at various things on the farm, but we are not a consulting service,” Patton said. “They (LGMA) take care of the compliance end, with the exception that if we find an immediate public health risk, we would contact the Department of Health.”
Once the CDFA auditors finish their ground-level work, the information is reviewed by LGMA for potential citations and follow-up training.
“They go out and make observations in the field with the handlers on paperwork and things and they send me their observations, and I determine whether their observations are in line with the metrics,” explained Jonathan Field, LGMA’s compliance officer.
Field can elevate an audit finding from a “Minor Infraction” to a “Minor Deviation” when a problem indicates that specific training is needed. He gave the example of when the auditors show up and the workers have pencils, gloves, or cell phones in their pockets, things which are typically not allowed when a crew is in the field harvesting produce.

“They find an employee with pencils in his pockets, so food safety comes and takes the pencils out and it’s done,” he said. “But why did that person have pencils in his pockets? Who was responsible for checking crews to make sure they don’t have pencils in their pockets? Are they being trained?”
Field said that a more effective approach would be to stop the harvest, call the crew together, and note that the company’s standard operating procedures say that no employee can have anything in their pockets while working in the field.
“Then, in a week or a few days later, when the crews come to harvest, the food safety person comes out and they do training about what is allowed in the field and in the pockets. Then there is a document available with the signatures of all the employees who took the training,” he said.
If they choose, LGMA handlers can adopt higher safety standards for their operation than the established metrics, Field said, but that means they are responsible for meeting their own higher standards during an audit.
“The metrics are the standard by which the industry is measured, so you have to adhere to those standards,” he said. “Your company can be higher, but when the auditors go out and check levels, you could be OK with the metrics but be out of compliance with your own standard operating procedure.”
LGMA recently produced a video of Field explaining how the group’s inspection and compliance program works.
Four citation categories Possible LGMA citations are grouped into these four categories:
- Flagrant Violation: A violation that significantly increased the risk of delivering unsafe product into commerce. Penalties can range from temporary to permanent decertification.
- Major Deviation: Does not necessarily result in unsafe product. Corrective action must be provided within five days. This citation can be elevated to the Flagrant level if not corrected.
- Minor Deviation: Does not necessarily result in unsafe product. Corrective action must be provided within five days. Multiple violations of the same type within a year may raise the citation level to a Major Deviation.
- Minor Infraction: Does not necessarily increase the risk of unsafe product. Corrective action can be taken before the auditor leaves the premises. Multiple violations will lead to a Minor Deviation.
The 2015-2016 annual report indicates there were no Flagrant Violations cited during that period, although there were 13 Major Deviations, 239 Minor Deviations, and 133 Minor Infractions.
One Major Deviation in the Environmental Assessment category involved a field close to an equestrian center. According to the annual report, the LGMA handler worked with the owners to make sure that any manure on their premises was moved to an appropriate distance from the leafy greens field.

In the Field Observations category, a Major Deviation was noted because a worker was seen eating lettuce in a part of a field under active harvest. The report states that the worker was reprimanded and temporarily suspended, the crew retrained on the issue, and the company foreman instructed to remind the crew prior to each day’s harvest that eating in the field is not allowed.
LGMA’s enhanced focus on training is designed to make sure the group’s members don’t become complacent, particularly about less serious infractions.
As stated on the LGMA website, “So rather than allowing minor corrections to continue being made in the field during an audit, we wanted handlers to go back and train people not to make these same mistakes again.
“We know that training can make a big difference in how leafy greens workers comply with required food safety practices as it helps ingrain the proper procedures into their everyday work habits.”
Complying with FSMA Field called California’s food safety programs “probably a step ahead from the rest of the country” due to the focus on training and compliance and ongoing efforts to encourage employees to take responsibility for helping to provide safe products.
“LGMA has made a very strong effort to work with FSMA and with the government to make sure that we were going to comply with what they needed,” he said. “The leafy greens industry in California is very confident that they will comply with FSMA and, in most cases, exceed the requirements of FSMA.”
From his state agency-level perspective, Patton said he is in “complete agreement” with that assessment.
“They are head and shoulders above other industries simply because they put themselves there,” he said. “LGMA has done a great job of getting information and training out to their members, and it’s working. There really has been a success story.”
The California Leafy Greens Products Handler Marketing Agreement was formed one year after the 2006 E. coli outbreak linked to Dole bagged baby spinach produced in the Salinas Valley. According to the U.S. Centers for Disease Control and Prevention, 199 people from 26 states were infected with the outbreak strain, 102 were hospitalized, 31 developed hemolytic uremic syndrome, and three people died.
(To sign up for a free subscription to Food Safety News, click here.)
]]> In a statement issued Wednesday, the Michigan Department of Health and Human Services noted that the 85 Genesee County cases compares with 20 shigellosis cases reported there in 2015 and four cases in 2014. Shigellosis is a highly contagious disease caused by four different strains of Shigella bacteria. Even a microscopic amount of contaminated fecal matter in food or water can cause infection if consumed. Most people infected with the bacteria develop diarrhea, fever and stomach cramps within a day or two after being exposed. The symptoms usually resolve within five to seven days. Some people who are infected may have no symptoms at all, but may still pass the Shigella bacteria to others. The spread of Shigella can be stopped by frequent and careful hand-washing with soap and by taking other hygiene measures. Flint’s water contamination problems began in April 2014 when the city switched its source from treated Lake Huron and Detroit River water to the Flint River. Because officials did not add corrosion inhibitors to the highly corrosive river water, it caused lead from aging pipes to leach into the city’s water supply. Between June 2014 and November 2015, there were 87 cases of infection with Legionella bacteria reported in Genesee County. Ten of those people died. However, no direct link was made between the spike in Legionnaires’ disease and the change in the water system. Some public health officials are speculating that the current shigellosis problem stems from Flint residents being afraid to use the tainted city water to wash their hands, even though hand-washing is a recommended method for limiting foodborne and other illnesses. According to Jim Henry, environmental health supervisor for Genesee County, “People aren’t bathing because they’re scared. Some people have mentioned that they’re not going to expose their children to the water again.” In an interview with CNN, Henry advised county residents not to rely on baby wipes, handed out for free at bottled water distribution centers, because they aren’t chlorinated, don’t kill the bacteria, and can’t replace thorough hand-washing. However, others find no connection between the water problems and the uptick in shigellosis cases. “We don’t know the exact reason,” said Dr. Gary Johnson, medical director at the Genesee County Health Department, in a Facebook post. “There isn’t a particular reason why.”
In a statement issued Wednesday, the Michigan Department of Health and Human Services noted that the 85 Genesee County cases compares with 20 shigellosis cases reported there in 2015 and four cases in 2014. Shigellosis is a highly contagious disease caused by four different strains of Shigella bacteria. Even a microscopic amount of contaminated fecal matter in food or water can cause infection if consumed. Most people infected with the bacteria develop diarrhea, fever and stomach cramps within a day or two after being exposed. The symptoms usually resolve within five to seven days. Some people who are infected may have no symptoms at all, but may still pass the Shigella bacteria to others. The spread of Shigella can be stopped by frequent and careful hand-washing with soap and by taking other hygiene measures. Flint’s water contamination problems began in April 2014 when the city switched its source from treated Lake Huron and Detroit River water to the Flint River. Because officials did not add corrosion inhibitors to the highly corrosive river water, it caused lead from aging pipes to leach into the city’s water supply. Between June 2014 and November 2015, there were 87 cases of infection with Legionella bacteria reported in Genesee County. Ten of those people died. However, no direct link was made between the spike in Legionnaires’ disease and the change in the water system. Some public health officials are speculating that the current shigellosis problem stems from Flint residents being afraid to use the tainted city water to wash their hands, even though hand-washing is a recommended method for limiting foodborne and other illnesses. According to Jim Henry, environmental health supervisor for Genesee County, “People aren’t bathing because they’re scared. Some people have mentioned that they’re not going to expose their children to the water again.” In an interview with CNN, Henry advised county residents not to rely on baby wipes, handed out for free at bottled water distribution centers, because they aren’t chlorinated, don’t kill the bacteria, and can’t replace thorough hand-washing. However, others find no connection between the water problems and the uptick in shigellosis cases. “We don’t know the exact reason,” said Dr. Gary Johnson, medical director at the Genesee County Health Department, in a Facebook post. “There isn’t a particular reason why.”
(To sign up for a free subscription to Food Safety News, click here.)
]]>

(To sign up for a free subscription to Food Safety News, click here.)
]]> The 134 people who became infected in the U.S. are from: Arizona, Colorado, Connecticut, Florida, Georgia, Illinois, Indiana, Iowa, Maine, Massachusetts, Michigan, Minnesota, Missouri, Nebraska, New Hampshire, New Jersey, New York, North Carolina, Ohio, South Dakota, Tennessee, Texas, Virginia, Washington and Wisconsin. Since April 2015, FDA has been detaining fresh cilantro from the Puebla region of Mexico after it was implicated in several foodborne illness outbreaks in the U.S. FDA issued an Import Alert on Aug. 27, 2015, announcing that it would detain fresh cilantro on a seasonal basis after joint U.S.-Mexico inspections from 2013-2015 at Puebla-area farms and packinghouses revealed “objectionable conditions” that could contaminate produce with “human fecal pathogens.” “Beginning in 2015, from April 1 through August 31, cilantro from this region has been and continues to be detained without physical examination at the U.S.-Mexican border and refused admission into the United States. The April through August time period aligns with the seasonality of previous C. cayetanensis outbreaks,” FDA noted in a Sept. 26 update on the situation. An exception is granted for fresh cilantro from 10 Puebla-area firms on a so-called “Green List.” Inclusion on the list means that the exporting companies have met certain good production practices criteria for exclusion from detention without physical examination under the Import Alert. FDA and two agencies of the Mexican government — the National Agro-Alimentary Health, Safety and Quality Service and Federal Commission for the Protection from Sanitary Risks — established production criteria to promote the safety of fresh and minimally processed agricultural products. They signed a statement of intent on July 24, 2014, to cooperate on a Produce Safety Partnership. This year was the first full season that FDA’s Import Alert for fresh cilantro from Puebla has been in effect. The agency also started testing cilantro from Puebla and, along with CDC, has been reaching out to industry on strategies to control and prevent Cyclospora. As of Sept. 14, 2015, CDC reported 546 cases of confirmed Cyclospora infection from 31 states. Of that total, 319 had experienced illness onset on or after May 1 of last year and had no history of international travel within two weeks before becoming sickened. Twenty-one people were hospitalized, but no deaths were reported. Illness clusters linked to restaurants or events were identified in Texas, Wisconsin and Georgia, and investigations in Wisconsin and Texas preliminarily identified cilantro as a “suspect vehicle,” according to CDC.
The 134 people who became infected in the U.S. are from: Arizona, Colorado, Connecticut, Florida, Georgia, Illinois, Indiana, Iowa, Maine, Massachusetts, Michigan, Minnesota, Missouri, Nebraska, New Hampshire, New Jersey, New York, North Carolina, Ohio, South Dakota, Tennessee, Texas, Virginia, Washington and Wisconsin. Since April 2015, FDA has been detaining fresh cilantro from the Puebla region of Mexico after it was implicated in several foodborne illness outbreaks in the U.S. FDA issued an Import Alert on Aug. 27, 2015, announcing that it would detain fresh cilantro on a seasonal basis after joint U.S.-Mexico inspections from 2013-2015 at Puebla-area farms and packinghouses revealed “objectionable conditions” that could contaminate produce with “human fecal pathogens.” “Beginning in 2015, from April 1 through August 31, cilantro from this region has been and continues to be detained without physical examination at the U.S.-Mexican border and refused admission into the United States. The April through August time period aligns with the seasonality of previous C. cayetanensis outbreaks,” FDA noted in a Sept. 26 update on the situation. An exception is granted for fresh cilantro from 10 Puebla-area firms on a so-called “Green List.” Inclusion on the list means that the exporting companies have met certain good production practices criteria for exclusion from detention without physical examination under the Import Alert. FDA and two agencies of the Mexican government — the National Agro-Alimentary Health, Safety and Quality Service and Federal Commission for the Protection from Sanitary Risks — established production criteria to promote the safety of fresh and minimally processed agricultural products. They signed a statement of intent on July 24, 2014, to cooperate on a Produce Safety Partnership. This year was the first full season that FDA’s Import Alert for fresh cilantro from Puebla has been in effect. The agency also started testing cilantro from Puebla and, along with CDC, has been reaching out to industry on strategies to control and prevent Cyclospora. As of Sept. 14, 2015, CDC reported 546 cases of confirmed Cyclospora infection from 31 states. Of that total, 319 had experienced illness onset on or after May 1 of last year and had no history of international travel within two weeks before becoming sickened. Twenty-one people were hospitalized, but no deaths were reported. Illness clusters linked to restaurants or events were identified in Texas, Wisconsin and Georgia, and investigations in Wisconsin and Texas preliminarily identified cilantro as a “suspect vehicle,” according to CDC. 
(To sign up for a free subscription to Food Safety News, click here.)
]]>The culprit was identified as Amanita pantherina, or Panther cap mushroom, which a worker reportedly gave the children to try Sept. 21 while guiding them through the garden at Micheltorena Elementary School in the Silver Lake neighborhood.

Officials with the Los Angeles Unified School District were not providing a specific number of illnesses linked to the mushroom due to differing reports. However, most of the sickened children were back in school on Wednesday, according to Monica Carazo, a school district spokesperson.
Principal Nichole Sakellarion held two meetings in the Micheltorena auditorium on Tuesday, one in the morning and one in the afternoon, to discuss the incident with parents and other concerned members of the public. Automated phone messages were also sent out to inform parents of what had happened.
District Superintendent Michelle King issued a statement Friday noting that the district’s environmental health and safety team is investigating the matter.
“In addition, in an abundance of caution, I have directed our safety team to inspect every District garden for potential hazards,” she said. There are more than 100 local school gardens in LA.
The Micheltorena community garden was installed in November 2010 to replace a parking lot on the corner of Sunset Boulevard and Micheltorena Street in Silver Lake. The garden had been hosting classes on Wednesdays during the school year and community potlucks on Thursday evenings, and it was open to the public on Saturdays.
Very few of the estimated 10,000 mushroom species in North America are considered poisonous, although proper identification is not an easy task. The consequences of ingesting one of the toxic variety can include gastrointestinal irritation, along with difficulty walking, speaking or breathing, and a decline in mental alertness. Some can even be fatal.
“The symptoms usually appear within 20 minutes to 4 hours of ingesting the mushrooms, and include nausea, vomiting, cramps, and diarrhea, which normally pass after the irritant has been flushed out by the body,” according to a LA school district safety alert issued Monday.
The school district recommends that people remove wild mushrooms found on school campuses and dispose of them in the trash and to closely supervise small children in areas where mushrooms are present.
Mycologists consider the Panther cap mushroom toxic but normally not deadly. It contains the psychoactive compounds ibotenic acid and muscimol. Because the species tends to grow beneath Monterey pines, it can often be found in urban parks in the San Francisco Bay area.
Experts advise teaching children not to eat wild mushrooms or plants while playing outdoors. Anyone who suspects that a child, or anyone, has eaten a wild mushroom is advised to call the national poison control number at 800-222-1222, contact a health care provider, or go to the nearest emergency department.
They also recommend collecting a sample of the mushroom the individual may have eaten and take it along to assist with identification.
The U.S. Centers for Disease Control and Prevention reported nine cases of mushroom poisoning in January 1997 in northern California involving the Amanita phalloides (death cap) mushroom species. All nine people were hospitalized, and two of them died.
(To sign up for a free subscription to Food Safety News, click here.)
]]>
(To sign up for a free subscription to Food Safety News, click here.)
]]> “The management team of Genki Sushi restaurants on Oahu and Kauai has given us their full cooperation, and the department is confident that they are in compliance with all health regulations,” said Health Director Dr. Virginia Pressler. Mary Hansen, chief administrative officer for Genki Sushi USA, said that the company’s employees scheduled to work in the Oahu and Kauai restaurants had been screened, tested, and vaccinated for Hepatitis A, and that all test results were negative. “It is very reassuring that none of our employees tested positive for the virus and we are happy that they can get back to work when the restaurants reopen. At the same time, our hearts go out to those who have the illness and hope for their speedy recovery,” she said. The state health department closed down the Genki Sushi restaurants on Oahu and Kauai on Aug. 16 after determining there was a link between the outbreak of Hepatitis A infections and frozen scallops served raw at the company’s outlets. The likely source of the contamination was identified as Sea Port Bay Scallops imported from the Philippines and distributed in Hawaii by Koha Oriental Foods and True World Foods. The U.S. Food and Drug Administration stated that laboratory analysis done Aug. 17 of two scallop samples collected on Aug. 11 tested positive for Hepatitis A. These samples were imported by Sea Port Products Corp. and were produced on Nov. 23 and 24, 2015, the agency stated. On Aug. 18, Sea Port Products Corp. recalled three lots of frozen bay scallops produced on Nov. 23 and 24, 2015. The lot numbers are 5885, 5886, and 5887. The products were distributed to California, Hawaii, and Nevada. The company indicated that the recalled products are not intended for retail sale. “The scallops received by True World Foods have not been distributed to any restaurants in the state, and were embargoed at their warehouse. The scallops served at Genki locations on the Big Island and Maui originated from a different supplier and have not been associated with the outbreak,” the health department noted in a statement posted on its website. According to news reports, Genki Sushi was Koha Oriental Food’s only client for those particular scallops, and the distributor has since changed sources.
“The management team of Genki Sushi restaurants on Oahu and Kauai has given us their full cooperation, and the department is confident that they are in compliance with all health regulations,” said Health Director Dr. Virginia Pressler. Mary Hansen, chief administrative officer for Genki Sushi USA, said that the company’s employees scheduled to work in the Oahu and Kauai restaurants had been screened, tested, and vaccinated for Hepatitis A, and that all test results were negative. “It is very reassuring that none of our employees tested positive for the virus and we are happy that they can get back to work when the restaurants reopen. At the same time, our hearts go out to those who have the illness and hope for their speedy recovery,” she said. The state health department closed down the Genki Sushi restaurants on Oahu and Kauai on Aug. 16 after determining there was a link between the outbreak of Hepatitis A infections and frozen scallops served raw at the company’s outlets. The likely source of the contamination was identified as Sea Port Bay Scallops imported from the Philippines and distributed in Hawaii by Koha Oriental Foods and True World Foods. The U.S. Food and Drug Administration stated that laboratory analysis done Aug. 17 of two scallop samples collected on Aug. 11 tested positive for Hepatitis A. These samples were imported by Sea Port Products Corp. and were produced on Nov. 23 and 24, 2015, the agency stated. On Aug. 18, Sea Port Products Corp. recalled three lots of frozen bay scallops produced on Nov. 23 and 24, 2015. The lot numbers are 5885, 5886, and 5887. The products were distributed to California, Hawaii, and Nevada. The company indicated that the recalled products are not intended for retail sale. “The scallops received by True World Foods have not been distributed to any restaurants in the state, and were embargoed at their warehouse. The scallops served at Genki locations on the Big Island and Maui originated from a different supplier and have not been associated with the outbreak,” the health department noted in a statement posted on its website. According to news reports, Genki Sushi was Koha Oriental Food’s only client for those particular scallops, and the distributor has since changed sources.  The source of the implicated scallops, De Oro Resources Inc., has been temporarily suspended by the government of the Philippines from distributing its products and ordered to test its employees for Hepatitis A. However, an agriculture official with the Philippines government said that all 129 workers who shuck scallops and work in the company’s processing plant had tested negative for the virus. As of Sept. 7, the case count in Hawaii’s Hepatitis A outbreak stood at 252, with 66 hospitalizations. All of those sickened have been adults. The state health department stated that onset of illness has ranged from June 12 to Aug. 30, and that officials still expect to see additional cases given the long incubation period for Hepatitis A. Symptoms can show up anywhere from 15 to 50 days after exposure to the virus. Hepatitis A symptoms can include fever, fatigue, loss of appetite, abdominal discomfort, dark urine, diarrhea, yellow skin and eyes, pale-colored stools, vomiting, and headaches and/or body aches. Hepatitis A vaccine or immune globulin injections are effective after exposure, but only if they are given within two weeks of exposure. Anyone who believes they have been exposed to the Hepatitis A virus should consult a health care provider.
The source of the implicated scallops, De Oro Resources Inc., has been temporarily suspended by the government of the Philippines from distributing its products and ordered to test its employees for Hepatitis A. However, an agriculture official with the Philippines government said that all 129 workers who shuck scallops and work in the company’s processing plant had tested negative for the virus. As of Sept. 7, the case count in Hawaii’s Hepatitis A outbreak stood at 252, with 66 hospitalizations. All of those sickened have been adults. The state health department stated that onset of illness has ranged from June 12 to Aug. 30, and that officials still expect to see additional cases given the long incubation period for Hepatitis A. Symptoms can show up anywhere from 15 to 50 days after exposure to the virus. Hepatitis A symptoms can include fever, fatigue, loss of appetite, abdominal discomfort, dark urine, diarrhea, yellow skin and eyes, pale-colored stools, vomiting, and headaches and/or body aches. Hepatitis A vaccine or immune globulin injections are effective after exposure, but only if they are given within two weeks of exposure. Anyone who believes they have been exposed to the Hepatitis A virus should consult a health care provider.
(To sign up for a free subscription to Food Safety News, click here.)
]]>Foodborne illnesses and food recalls seem to dominate the news these days. All across the globe, we hear about people being sickened by contaminated milk, flour, fresh produce, raw seafood, frozen entrées, and myriad other products.
Some food-related recalls involve only one product lot and no reported illnesses. Others involve thousands of pounds of food sold nationwide, with hundreds sickened, dozens hospitalized, and occasionally, some fatalities.
According to the U.S. Centers for Disease Control and Prevention (CDC), about 48 million people become ill each year from a foodborne pathogen acquired in this country. Of those, about 128,000 are hospitalized and 3,000 die.
CDC has linked about 9.4 million episodes of foodborne illnesses each year to 31 identifiable pathogens, although more than 95 percent of these related illnesses, hospitalizations and deaths are caused by only 15 of them.
Because many foodborne illnesses are not reported, and sometimes even when they are, the cause for 80 percent of the ones acquired in the U.S. is never identified.
Besides the individual, personal costs, the economic costs from medical expenses and lost productivity are staggering. The U.S. Department of Agriculture’s Economic Research Service estimated the financial burden of the 15 leading foodborne illnesses acquired in the U.S. at about $15.5 billion in 2013 dollars.
Last year, a study from Ohio State University put a much higher price tag — and a wide-ranging estimate — on the total cost of foodborne illness in the U.S.: $55.5 to $93.2 billion per year.
Then there are the quantifiable costs to the recalling businesses. A 2011 Grocery Manufacturers Association survey of 34 international firms found that more than half of them had been affected by a food recall in the previous five years. Of those, 18 percent reported lost sales of $30 million to $99 million, and 5 percent lost $100 million or more.
A global supply chain
The reasons driving this situation are as complex as today’s food supply chain, which stretches across continents. As our food supply becomes more globalized, so do the chances for problems, and the increasing complexity created by globalization makes it even tougher to find and eliminate those problems.
 As the world’s population becomes more centralized in cities and increasingly less dispersed on farms, reliance on others for our food supply will become the norm, states a 2015 report by Zurich-based Swiss Re entitled, “Food Safety in a Globalised World.”
As the world’s population becomes more centralized in cities and increasingly less dispersed on farms, reliance on others for our food supply will become the norm, states a 2015 report by Zurich-based Swiss Re entitled, “Food Safety in a Globalised World.”
“Assuring this supply today is not possible without globalisation. It is a process involving countless partners and interfaces, requiring logistics that are complicated and are growing more so by the day,” the group’s report states. “But where there is complication there may be error. And error may lead to harm.”
As writer Beth Kowitt put it in a May 6, 2016, Fortune Magazine article, our pantry is global and so are the chances for contamination. Americans want certain food products available year-round and we want them cheap.
We bring in strawberries from Mexico in the winter, for example, and some exporting countries have different food safety standards and inspection regimes, Kowitt notes.
“The global supply chain has not only given us a cornucopia of food choices; it has also cut costs. The downside is that it has made preventing food-borne contamination nearly impossible. By the time milk tainted with melamine produced in China was detected, it had already been exported to 47 countries by way of milk-containing products,” she writes.
The question of consumer trust
For industry, the cost of recalls inevitably impacts the bottom line. However, the impact to customer trust in a given brand name is harder to quantify.
No food manufacturer wants a consumer to hesitate about buying their product, to stand there in the grocery store, checking the label, and wondering whether the item has been safely produced and whether eating it might harm them or their family members.
Naturally consumers want to know which products are safe and where to find information they can trust. However, according to a March 2016 analysis from Sullivan, Higdon & Sink, an ad agency based in Kansas City, MO, only about one-third of American consumers trust food companies to give them accurate product information.
The good news is that the level of trust has gone up in recent years, SHS notes, mainly due to better and more concerted efforts at consumer education to fill the gap. After all, consumers vote with their wallets (and, increasingly, share their sentiments on social media sites), so any erosion of trust will be felt quickly and painfully and can take a long time to shore back up.
Key to understanding this is the elusive concept of having a “social license to operate.” As explained in a Sept. 6, 2016, Food Safety magazine piece, this means “… the ability for any organization to operate with the confidence of stakeholders and that its activities are deemed morally and socially legitimate. Companies are judged based on a set of values collectively shared across the continuum of food system stakeholders.”
When contamination is discovered, people get sick, or recalls are announced, there is a perceived breach of this social license. The fragile trust between producers and consumers is consequently damaged and only restored (if at all) by being laboriously earned back bit by bit over time.
Or, as the magazine article put it, “The very essence of social license is transparency, inclusiveness and informed consent. Whether we agree with the concept of food social license or not, or even think that it has always been there, we now have an opportunity for stakeholders to make the food industry more democratic, more accessible and relevant to the broader public.”
Diversity, globalization, supply chain complexity and fragile consumer trust have collided to present a perfect storm of challenges for today’s food industry. How companies adjust their operations to handle technology, traceability, and new federal regulations, among other management decisions, will determine how well they weather those challenges.
(To sign up for a free subscription to Food Safety News, click here.)
]]>
 “We can confirm that that the department is investigating the incident at Pablo’s Mexican Cuisine and DBPR conducted an environmental assessment, which the [sic] can find on their website. We will have more information when our investigation concludes,” Brad Dalton, deputy press secretary for the Florida Department of Health, said in an email Wednesday afternoon. Dalton was referring to the Florida Department of Business & Professional Regulation, which recently inspected the restaurant at 12 N. 2nd St. in Fernandina Beach, a city on Amelia Island off Florida’s northeastern coast. The inspections, done Aug. 19, 23, 24 and 25, revealed a number of “basic,” “intermediate,” and “high priority” food safety problems. These ranged from temperature abuses and mold to flying insects in the bar and signs of roach activity. The inspection reports show that either the violations were corrected at the time the inspections were done, warnings were given, or the timeline was extended for compliance with applicable regulations. The most recent report from the Aug. 25 inspection indicates that the restaurant “met inspection standards.”
“We can confirm that that the department is investigating the incident at Pablo’s Mexican Cuisine and DBPR conducted an environmental assessment, which the [sic] can find on their website. We will have more information when our investigation concludes,” Brad Dalton, deputy press secretary for the Florida Department of Health, said in an email Wednesday afternoon. Dalton was referring to the Florida Department of Business & Professional Regulation, which recently inspected the restaurant at 12 N. 2nd St. in Fernandina Beach, a city on Amelia Island off Florida’s northeastern coast. The inspections, done Aug. 19, 23, 24 and 25, revealed a number of “basic,” “intermediate,” and “high priority” food safety problems. These ranged from temperature abuses and mold to flying insects in the bar and signs of roach activity. The inspection reports show that either the violations were corrected at the time the inspections were done, warnings were given, or the timeline was extended for compliance with applicable regulations. The most recent report from the Aug. 25 inspection indicates that the restaurant “met inspection standards.”  Pablo’s was open on Wednesday, and an employee there told Food Safety News that the restaurant had not been closed either during the recent inspections or afterward. “It’s weird because all the employees eat here. My own family eats here, and none of us has gotten sick ever,” she said. “It’s very strange and out of nowhere.” Salmonella bacteria usually cause diarrhea, fever, and abdominal cramps 12 to 72 hours after infection. The illness typically lasts four to seven days, and most people recover without treatment. However, diarrhea can sometimes be so severe that the patient needs to be hospitalized. The infection may spread from the intestines to the bloodstream and then to other body sites. In these cases, Salmonella can cause death unless the person is treated promptly with antibiotics. The elderly, infants, and those with impaired immune systems are more likely to have a severe illness.
Pablo’s was open on Wednesday, and an employee there told Food Safety News that the restaurant had not been closed either during the recent inspections or afterward. “It’s weird because all the employees eat here. My own family eats here, and none of us has gotten sick ever,” she said. “It’s very strange and out of nowhere.” Salmonella bacteria usually cause diarrhea, fever, and abdominal cramps 12 to 72 hours after infection. The illness typically lasts four to seven days, and most people recover without treatment. However, diarrhea can sometimes be so severe that the patient needs to be hospitalized. The infection may spread from the intestines to the bloodstream and then to other body sites. In these cases, Salmonella can cause death unless the person is treated promptly with antibiotics. The elderly, infants, and those with impaired immune systems are more likely to have a severe illness.
(To sign up for a free subscription to Food Safety News, click here.)
]]> A raw milk sample collected at Heber Valley Milk by a Utah Department of Agriculture and Food inspector tested positive for Salmonella Saintpaul on Aug. 23, the health department stated. However, the most recent tests showed no signs of Salmonella and the dairy has been allowed to resume sales. Grant Kohler, owner of Heber Valley Milk, said he is working with state officials to try to figure out what happened. “We don’t know what the cause was and we’re not 100 percent sure that it’s our place,” he said. “We will do whatever we need to do to make sure we’re selling a safe product.”
A raw milk sample collected at Heber Valley Milk by a Utah Department of Agriculture and Food inspector tested positive for Salmonella Saintpaul on Aug. 23, the health department stated. However, the most recent tests showed no signs of Salmonella and the dairy has been allowed to resume sales. Grant Kohler, owner of Heber Valley Milk, said he is working with state officials to try to figure out what happened. “We don’t know what the cause was and we’re not 100 percent sure that it’s our place,” he said. “We will do whatever we need to do to make sure we’re selling a safe product.”
 Raw milk comes from cows, goats or sheep and has not been pasteurized to kill harmful bacteria. This raw, unpasteurized milk can contain dangerous bacteria such as Campylobacter, Salmonella, Listeria and E. coli, which can all cause foodborne illnesses. Raw milk contaminated with disease-causing bacteria does not smell or look any different from uncontaminated raw milk, and there is no easy way for the consumer to know whether raw milk is contaminated. According to the Utah health department, since 2009 there have been 30 documented outbreaks associated with raw milk sold at dairies statewide, with more than 400 people becoming ill. Utah allows on-farm sales of raw milk as long as the milk producer owns the store. Monthly testing for bacteria and pathogens is required, and animals must be tested before production and every six months thereafter. The bottles must be labeled as raw milk and must carry a warning label. Public health officials warn that drinking raw milk may be dangerous, regardless where it is obtained. Raw milk should not be consumed by young children, pregnant women, the elderly, people with weakened or compromised immune systems, or anyone who does not want to become ill. Both the health department and the dairy emphasized that those who choose to drink raw milk should follow these steps to reduce the risk of illness:
Raw milk comes from cows, goats or sheep and has not been pasteurized to kill harmful bacteria. This raw, unpasteurized milk can contain dangerous bacteria such as Campylobacter, Salmonella, Listeria and E. coli, which can all cause foodborne illnesses. Raw milk contaminated with disease-causing bacteria does not smell or look any different from uncontaminated raw milk, and there is no easy way for the consumer to know whether raw milk is contaminated. According to the Utah health department, since 2009 there have been 30 documented outbreaks associated with raw milk sold at dairies statewide, with more than 400 people becoming ill. Utah allows on-farm sales of raw milk as long as the milk producer owns the store. Monthly testing for bacteria and pathogens is required, and animals must be tested before production and every six months thereafter. The bottles must be labeled as raw milk and must carry a warning label. Public health officials warn that drinking raw milk may be dangerous, regardless where it is obtained. Raw milk should not be consumed by young children, pregnant women, the elderly, people with weakened or compromised immune systems, or anyone who does not want to become ill. Both the health department and the dairy emphasized that those who choose to drink raw milk should follow these steps to reduce the risk of illness:
- Only buy raw milk from stores or dairies permitted by law to sell it. However, a government permit does not guarantee that raw milk will be free from disease-causing bacteria.
- Keep raw milk and raw milk products refrigerated at or below 40 degrees F.
- Transport milk from the store to your home in a cooler with ice packs.
- Do not let raw milk sit out at room temperature.
(To sign up for a free subscription to Food Safety News, click here.)
 This trend hasn’t been lost on Rex Lawrence, who recently launched Joe Food Safety, a site designed to connect food safety jobs with job seekers.
This trend hasn’t been lost on Rex Lawrence, who recently launched Joe Food Safety, a site designed to connect food safety jobs with job seekers.
“On the recruiting side, we are always looking at food safety. Things like FSMA, what’s happening within the industry. It’s heating up and it’s not going to go away. I don’t believe it to be kind of a flash in the pan. I think it’s going to do nothing but get bigger and better, and it’s going to need good people,” Lawrence said.
He said the online service, initially free to those looking to fill food safety positions, will include help writing job titles and descriptions and then filtering resumes as they come in.
“If you’re a company and you’re looking for a director of food safety, we make sure you aren’t getting a resume from a kid who’s flipping burgers at McDonald’s, let’s say,” Lawrence said.
After working for a produce company, Lawrence noticed that it was hard to fill food safety director positions. That experience led him to set up Joe Produce in 2012, which focuses on produce industry employment, and then to connect with educational and other settings where students may be entering food safety fields.
“With the heavier and overall focus on food safety and more employment in food safety, I think the money flowing into food safety is going to increase. There’s going to be more studies and more food safety research,” he said.
 Universities and other training grounds are paying attention. Michigan State University in East Lansing launched an online Master of Science in Food Safety program in 2002 after a study revealed “an undeniable need on the part of the food industry, government, and public health for their employees to be specifically educated in the many aspects of safeguarding our food supply.”
Universities and other training grounds are paying attention. Michigan State University in East Lansing launched an online Master of Science in Food Safety program in 2002 after a study revealed “an undeniable need on the part of the food industry, government, and public health for their employees to be specifically educated in the many aspects of safeguarding our food supply.”
MSU’s curriculum is designed to permit U.S.-based or international students to earn their master’s degree in food safety while continuing to work. Program statistics indicate there were about 400 graduates between 2002 and 2012.
North Carolina State University in Raleigh offers bachelor’s, master’s and doctoral programs in science, with options to focus on food, nutrition, or bioprocessing.
“The Bureau of Labor and Statistics says that job growth for food related occupations are expected to be driven by the demand for new food products and food safety measures. Food research is expected to increase because the public is more aware of nutrition, health, food safety, and the need to keep herd animals from getting infections,” according to NC State’s program site.
Food safety-related certifications are in increasing demand, with an alphabet soup of acronyms showing up in job ads. Employers, whether farms, food processors, national distributors or international exporters, often want to see GAP, GMP, HACCP, GFSI and other specific experience and/or certifications on an applicant’s resume. There are also CFP, SQF, BRC, IFS, BAP and FSSC 22000 certifications out there to up the ante.
 Helping to fill that need is the National Registry of Food Safety Professionals in Orlando, FL, which develops and maintains certification examination programs for food safety. It tests for food safety manager and HACCP certifications and offers a variety of ways to take the exams.
Helping to fill that need is the National Registry of Food Safety Professionals in Orlando, FL, which develops and maintains certification examination programs for food safety. It tests for food safety manager and HACCP certifications and offers a variety of ways to take the exams.
Those who earn food safety-related degrees and certifications can expect to pull in initial salaries ranging from about $37,000 to about $63,000 per year, according to federal government estimates. Where an individual salary lands along that range depends on whether the focus is on agriculture or food science, food service management, or some other related work.
Training is becoming more important in the food industry as companies realize they can’t just assign food safety duties to an administrative staffer who may not have sufficient background to do the job. The stakes are high since a serious recall could put a company out of business, and brand loyalty is hard to win back once it’s been tarnished.
Lawrence said that in the past big companies were usually the ones pushing for more training and certifications from employees because they had so much to lose. However, he said that smaller firms, including packers, producers and manufacturers, are now seeing the need.
“Companies are realizing this is serious,” he said. “Just to do business, they need someone with food safety certification and auditing and a certain level of certification. The smaller and mid-sized companies now are stepping up to the plate because they want to do business with the Costcos and the Wal-Marts of the world.”
(To sign up for a free subscription to Food Safety News, click here.)
]]> “The evidence demonstrates that L. monocytogenes has maintained its presence within your production facility since 2015. The reoccurring presence of an identical strain of L. monocytogenes in your environment indicates a resident strain or niche harborage site present in the facility,” according to the letter. “These findings also demonstrate that your sanitation procedures have historically been inadequate to control, reduce, or eliminate this pathogenic organism from your facility. It is essential to identify the areas of the food processing plant where this organism is able to grow and survive and to take such corrective actions as necessary to eradicate the organism by rendering these areas unable to support the growth and survival of the organism,” FDA told the company. Jeni’s has been using its Columbus facility to make frozen flavor bases after Listeria was found last year in its production kitchen and in the ice cream sample. Since then, the company has had its finished ice creams produced by Smith Dairy in Orrville, OH. The contamination was initially found in a pint of Jeni’s dark chocolate ice cream randomly tested in Nebraska. The company then halted production, recalled all of its products, and embarked on a thorough cleaning, sanitizing and reconfiguring of its production kitchen that reportedly cost $200,000. Jeni’s also destroyed 265 tons of ice cream worth more than $2.5 million. FDA inspectors who visited Jeni’s production facility in Columbus eight times in April 2015 reported inadequate pathogen controls and cleanliness procedures, which company president and CEO John Lowe indicated at the time had been fixed. Lowe did not respond to a request for comment Wednesday from Food Safety News. However, the company sent out a statement from Mary Kamm, its quality and new product introduction leader, noting that it was the only comment Jeni’s would make about the FDA warning letter. Kamm stressed that a “good Listeria control program” works by constantly searching for the bacteria and then eradicating it before it has a chance to spread to any food contact surfaces. “We believe our final product testing to be the most thorough in the nation,” she said in the statement. “As a result of our sanitation and other food safety procedures, our environmental testing program and our test and hold procedures, we can assure everyone that the food we produce is absolutely 100% safe.”
“The evidence demonstrates that L. monocytogenes has maintained its presence within your production facility since 2015. The reoccurring presence of an identical strain of L. monocytogenes in your environment indicates a resident strain or niche harborage site present in the facility,” according to the letter. “These findings also demonstrate that your sanitation procedures have historically been inadequate to control, reduce, or eliminate this pathogenic organism from your facility. It is essential to identify the areas of the food processing plant where this organism is able to grow and survive and to take such corrective actions as necessary to eradicate the organism by rendering these areas unable to support the growth and survival of the organism,” FDA told the company. Jeni’s has been using its Columbus facility to make frozen flavor bases after Listeria was found last year in its production kitchen and in the ice cream sample. Since then, the company has had its finished ice creams produced by Smith Dairy in Orrville, OH. The contamination was initially found in a pint of Jeni’s dark chocolate ice cream randomly tested in Nebraska. The company then halted production, recalled all of its products, and embarked on a thorough cleaning, sanitizing and reconfiguring of its production kitchen that reportedly cost $200,000. Jeni’s also destroyed 265 tons of ice cream worth more than $2.5 million. FDA inspectors who visited Jeni’s production facility in Columbus eight times in April 2015 reported inadequate pathogen controls and cleanliness procedures, which company president and CEO John Lowe indicated at the time had been fixed. Lowe did not respond to a request for comment Wednesday from Food Safety News. However, the company sent out a statement from Mary Kamm, its quality and new product introduction leader, noting that it was the only comment Jeni’s would make about the FDA warning letter. Kamm stressed that a “good Listeria control program” works by constantly searching for the bacteria and then eradicating it before it has a chance to spread to any food contact surfaces. “We believe our final product testing to be the most thorough in the nation,” she said in the statement. “As a result of our sanitation and other food safety procedures, our environmental testing program and our test and hold procedures, we can assure everyone that the food we produce is absolutely 100% safe.”  FDA acknowledged in the Aug. 9 warning letter the “extensive corrective actions” the company took last year after the positive Listeria results. Jeni’s hired a sanitation consultant and shut down production to deep clean and sanitize the facility. It also hired a food safety director.
FDA acknowledged in the Aug. 9 warning letter the “extensive corrective actions” the company took last year after the positive Listeria results. Jeni’s hired a sanitation consultant and shut down production to deep clean and sanitize the facility. It also hired a food safety director.
 The illnesses occurred following an annual reunion July 15-17 in Richey, MT, a town of about 200 people about 50 miles north of Glendive in the northeastern part of the state. Various food items served at the event were taken to the state lab in Helena for testing, but no specific food ingredient was identified as the source of the E. coli contamination. “We’ve narrowed it down to a meal, but due to a lack of samples and the way it was served, it wasn’t possible to identify the exact item,” Fladager said Wednesday. According to an Aug. 11 update from the Dawson County Health Department, which was posted on Facebook, all the individuals who became ill with comparable symptoms had attended an evening meal served July 16 at Richey High School. “Health officials are working with the caterer who provided the meal to determine how the meal became contaminated,” the Facebook post noted. The name of the caterer was not released. County health officials also thanked everyone who completed an online questionnaire and all the food vendors who cooperated with the investigation. As of Aug. 11, no additional E. coli cases had been reported, according to the department.
The illnesses occurred following an annual reunion July 15-17 in Richey, MT, a town of about 200 people about 50 miles north of Glendive in the northeastern part of the state. Various food items served at the event were taken to the state lab in Helena for testing, but no specific food ingredient was identified as the source of the E. coli contamination. “We’ve narrowed it down to a meal, but due to a lack of samples and the way it was served, it wasn’t possible to identify the exact item,” Fladager said Wednesday. According to an Aug. 11 update from the Dawson County Health Department, which was posted on Facebook, all the individuals who became ill with comparable symptoms had attended an evening meal served July 16 at Richey High School. “Health officials are working with the caterer who provided the meal to determine how the meal became contaminated,” the Facebook post noted. The name of the caterer was not released. County health officials also thanked everyone who completed an online questionnaire and all the food vendors who cooperated with the investigation. As of Aug. 11, no additional E. coli cases had been reported, according to the department.
(To sign up for a free subscription to Food Safety News, click here.)
]]>
 Nearly 100 people have been sickened in this outbreak so far, which began in June and is centered on Oahu, although there are victims who visited there but are living on neighbor islands. The department posted the dates of potential exposure at the Chili’s outlet at 590 Farrington Highway in Kapolei and from the Hawaiian Airlines flight list. State Epidemiologist Dr. Sarah Park reiterated that no infections have been linked to possible exposure at Chili’s or Hawaiian Airlines and that the two companies are not being considered sources of the outbreak. “We are alerting the public only as a precaution; the risk of transmission is extremely low and these businesses are working with us to help prevent potentially new cases in our community,” she said. The department noted that although it is not a food service establishment, Hawaiian Airlines has been named because the infected crew member served inflight food and beverages to passengers. The airline has posted information about the situation here and advised the public that it will reimburse “reasonable out-of-pocket expenses upon submission of documentation” for any customer on the affected flights who, after consulting a medical provider, needs screening and/or vaccination for Hepatitis A. The reimbursement request must be submitted by Sept. 30. “We have had no other reported cases among staff and are proactively screening all crew who worked with this crewmember before they return to duty,” the airline stated. Park cautioned that people exposed to the virus can transmit it even if they’re unaware they have it. “The most infectious period for this disease may be as much as two weeks before the onset of symptoms — before the individual even knows he or she is sick,” she said. The department has also identified confirmed Hepatitis A cases among workers at Baskin-Robbins, Cosco Bakery, Sushi Shiono and Taco Bell. All are on Oahu except for Sushi Shiono, which is north of Kailua-Kona on the Big Island of Hawaii. According to a Wednesday news report, 24 employees of Sushi Shiono were tested for Hepatitis A and all results were negative. The worker who tested positive is believed to have contracted the virus while on Oahu. Anyone who consumed food or beverage products from the named businesses during the identified periods may have been exposed to the disease, the health department stated. They are advised to:
Nearly 100 people have been sickened in this outbreak so far, which began in June and is centered on Oahu, although there are victims who visited there but are living on neighbor islands. The department posted the dates of potential exposure at the Chili’s outlet at 590 Farrington Highway in Kapolei and from the Hawaiian Airlines flight list. State Epidemiologist Dr. Sarah Park reiterated that no infections have been linked to possible exposure at Chili’s or Hawaiian Airlines and that the two companies are not being considered sources of the outbreak. “We are alerting the public only as a precaution; the risk of transmission is extremely low and these businesses are working with us to help prevent potentially new cases in our community,” she said. The department noted that although it is not a food service establishment, Hawaiian Airlines has been named because the infected crew member served inflight food and beverages to passengers. The airline has posted information about the situation here and advised the public that it will reimburse “reasonable out-of-pocket expenses upon submission of documentation” for any customer on the affected flights who, after consulting a medical provider, needs screening and/or vaccination for Hepatitis A. The reimbursement request must be submitted by Sept. 30. “We have had no other reported cases among staff and are proactively screening all crew who worked with this crewmember before they return to duty,” the airline stated. Park cautioned that people exposed to the virus can transmit it even if they’re unaware they have it. “The most infectious period for this disease may be as much as two weeks before the onset of symptoms — before the individual even knows he or she is sick,” she said. The department has also identified confirmed Hepatitis A cases among workers at Baskin-Robbins, Cosco Bakery, Sushi Shiono and Taco Bell. All are on Oahu except for Sushi Shiono, which is north of Kailua-Kona on the Big Island of Hawaii. According to a Wednesday news report, 24 employees of Sushi Shiono were tested for Hepatitis A and all results were negative. The worker who tested positive is believed to have contracted the virus while on Oahu. Anyone who consumed food or beverage products from the named businesses during the identified periods may have been exposed to the disease, the health department stated. They are advised to:
- Contact their healthcare providers about the possibility of receiving Hepatitis A vaccine or immune globulin (IG), which may provide some protection against the disease if administered within two weeks after exposure.
- Monitor their health for symptoms of Hepatitis A infection up to 50 days after exposure.
- Wash their hands frequently and thoroughly with soap and warm water, especially after using the bathroom or changing a diaper, and before preparing food.
- Stay at home and contact their healthcare provider immediately if symptoms of Hepatitis A infection develop.
The health department stated that to help prevent the spread of disease during the investigation, the public is encouraged to talk to their healthcare providers about vaccination. A statewide list of vaccinating pharmacies is available here, or people in Hawaii can call the Aloha United Way information and referral line at 2-1-1. Symptoms of Hepatitis A infection include fever, fatigue, loss of appetite, abdominal discomfort, dark urine, diarrhea, and yellow skin and eyes. Individuals, including food service employees, exhibiting symptoms of Hepatitis A should stay home and contact their healthcare provider. Additional information about Hepatitis A can be found here and here.
(To sign up for a free subscription to Food Safety News, click here.)
]]> The Oregon Potato Co., doing business as Freeze Pack, was told in a July 15 warning letter from FDA’s Seattle District Office that, following a March inspection of its processing plant in Pasco, WA, a number of environmental swabs taken there tested positive for the pathogen. FDA’s laboratory analysis of samples collected March 8 and 9 confirmed that 19 out 106 environmental swabs tested positive for Listeria monocytogenes. The warning letter noted that seven positive environmental swabs were collected from direct food contact surfaces in the plant’s processing and packaging rooms during the production of individually quick frozen (IQF) diced onions. Those direct food contact surface areas included:
The Oregon Potato Co., doing business as Freeze Pack, was told in a July 15 warning letter from FDA’s Seattle District Office that, following a March inspection of its processing plant in Pasco, WA, a number of environmental swabs taken there tested positive for the pathogen. FDA’s laboratory analysis of samples collected March 8 and 9 confirmed that 19 out 106 environmental swabs tested positive for Listeria monocytogenes. The warning letter noted that seven positive environmental swabs were collected from direct food contact surfaces in the plant’s processing and packaging rooms during the production of individually quick frozen (IQF) diced onions. Those direct food contact surface areas included:
- The chiller water and the interior north wall of the water chiller. Water from this chiller is not treated and is recirculated back to the blancher/chiller and used directly on blanched diced onions as a coolant;
- A white nylon strip in the tunnel discharge chute between the IQF freezer and the finished product packaging room. Blanched, finished onions are conveyed and come into direct contact with the nylon strip; and,
- The metal arm on the chain conveyor belt between the IQF freezer and packaging room where blanched, finished product is conveyed directly on this conveying system and comes into contact with the metal arm.
The remaining 12 positive environmental swabs were collected from locations in the plant’s processing room and the packaging room in areas adjacent to food contact surfaces and non-direct food contact surfaces, FDA stated. The 19 environmental samples were also analyzed using whole genome sequencing (WGS), which FDA noted can establish direct links between clinical isolates from sick people and food or environmental sources. The analysis found links between the isolates from the production plant and sick people, the agency stated. “The WGS phylogenetic analysis establishes that there are at least two different strains of L. monocytogenes present in the facility, with one strain containing 17 isolates and the second strain containing two isolates,” FDA stated.  “Specifically, the WGS analysis of the strain with 17 isolates showed that the isolates are identical to each other. WGS analysis of the strain with two isolates showed that the isolates are identical to eight cases of human illness dating back to 2013, and to six isolates from finished products. These finished products included onions, with two isolates in 2014, and green beans with three isolates in 2015, tested by a third party laboratory, and a single isolate from white sweet corn collected and tested by the state of Ohio in 2016. Additional investigation established that at least six individuals were hospitalized as a result of related L. monocytogenes associated illness,” according to the warning letter. Violations of Current Good Manufacturing Practice (CGMP) regulations were also observed at the facility, FDA told the company. These problems included accumulated food debris and grime on some food contact surfaces, insufficient measures to keep drip or condensate from contaminating food or food contact surfaces or food packaging materials, and inadequate ventilation or control equipment to minimize vapors — including steam — from contaminating food items. Oregon Potato Co. contacted FDA after receiving the warning letter and detailed corrective actions it was taking. The company also ceased production in order to clean and sanitize all surfaces in the facility, FDA noted, and submitted environmental testing samples taken alongside those taken by the agency, which were negative for Listeria. However, FDA responded that it was unable to evaluate the adequacy of the measures to be taken without further information and that there was no timeline submitted for completion or for resuming operations. As for the negative Listeria results the company submitted, FDA questioned the firm’s sample testing.
“Specifically, the WGS analysis of the strain with 17 isolates showed that the isolates are identical to each other. WGS analysis of the strain with two isolates showed that the isolates are identical to eight cases of human illness dating back to 2013, and to six isolates from finished products. These finished products included onions, with two isolates in 2014, and green beans with three isolates in 2015, tested by a third party laboratory, and a single isolate from white sweet corn collected and tested by the state of Ohio in 2016. Additional investigation established that at least six individuals were hospitalized as a result of related L. monocytogenes associated illness,” according to the warning letter. Violations of Current Good Manufacturing Practice (CGMP) regulations were also observed at the facility, FDA told the company. These problems included accumulated food debris and grime on some food contact surfaces, insufficient measures to keep drip or condensate from contaminating food or food contact surfaces or food packaging materials, and inadequate ventilation or control equipment to minimize vapors — including steam — from contaminating food items. Oregon Potato Co. contacted FDA after receiving the warning letter and detailed corrective actions it was taking. The company also ceased production in order to clean and sanitize all surfaces in the facility, FDA noted, and submitted environmental testing samples taken alongside those taken by the agency, which were negative for Listeria. However, FDA responded that it was unable to evaluate the adequacy of the measures to be taken without further information and that there was no timeline submitted for completion or for resuming operations. As for the negative Listeria results the company submitted, FDA questioned the firm’s sample testing.  “Given that FDA’s sampling revealed 19 environmental swabs that tested positive for L. monocytogenes, we are concerned with the adequacy of your sampling program. Your response included the general protocol reference guide for the method utilized by your laboratory. However, your firm did not provide detailed documents on the actual laboratory method that was performed to provide a comprehensive review of this issue,” the warning letter stated. The letter also mentioned the company’s recalled frozen vegetables:
“Given that FDA’s sampling revealed 19 environmental swabs that tested positive for L. monocytogenes, we are concerned with the adequacy of your sampling program. Your response included the general protocol reference guide for the method utilized by your laboratory. However, your firm did not provide detailed documents on the actual laboratory method that was performed to provide a comprehensive review of this issue,” the warning letter stated. The letter also mentioned the company’s recalled frozen vegetables:
(To sign up for a free subscription to Food Safety News, click here.)
 An all-class reunion held last month in Richey, MT, reportedly left some participants with more than pleasant memories. At least 30 people from eight states were sickened with E. coli O157 by an unknown food item, and several of them had to be hospitalized. “Currently more than 30 persons have been reported ill and at least 6 have been hospitalized. The event was attended by people from multiple states. At this time we have reports of cases in 8 states. In Montana 6 counties have been affected. New cases are still being reported and additional laboratory results are pending,” according to a July 28 Facebook post from the Dawson County Health Department in Glendive, MT. County health officials said those who became ill had attended the Richey Centennial and All-Class Reunion held July 15-17 in the small town in northeastern Montana. Officials have been interviewing people who attended one or more days of the event and have asked them to complete an online survey. The introduction to the online survey notes that more information will be released when it becomes available and that, in the interim, people are asked “to refrain from speculation regarding the cause” of the outbreak. News reports suggested that some of the food served during the reunion weekend may have been taken to people’s homes as leftovers. Meanwhile, a variety of food samples from the reunion are being tested and state and local public health officials are trying to track down where the E. coli bacteria may have come from. “While we are narrowing down the source, it is too early to tell what the exact cause of the outbreak was,” noted the county’s Facebook post. “Local food vendors have been very cooperative in this investigation. We encourage anyone who attended the event to not consume any leftover foods they may have brought back from the event and to contact their local health department if they still have leftover food, in case it may become of interest.” The Dawson County Health Department and the Montana Department of Public Health and Human Services are working together on the outbreak. Infection with E. coli bacteria often causes several stomach cramps and diarrhea (often bloody) and vomiting. Fever may be present, but in some cases, it is not. Symptoms usually appear within two to eight days after exposure. Most people get better within five to seven days. Some infections are very mild, but others are severe or even life-threatening, particularly if the person develops the kidney-related complication known as HUS (hemolytic uremic syndrome).
]]>
An all-class reunion held last month in Richey, MT, reportedly left some participants with more than pleasant memories. At least 30 people from eight states were sickened with E. coli O157 by an unknown food item, and several of them had to be hospitalized. “Currently more than 30 persons have been reported ill and at least 6 have been hospitalized. The event was attended by people from multiple states. At this time we have reports of cases in 8 states. In Montana 6 counties have been affected. New cases are still being reported and additional laboratory results are pending,” according to a July 28 Facebook post from the Dawson County Health Department in Glendive, MT. County health officials said those who became ill had attended the Richey Centennial and All-Class Reunion held July 15-17 in the small town in northeastern Montana. Officials have been interviewing people who attended one or more days of the event and have asked them to complete an online survey. The introduction to the online survey notes that more information will be released when it becomes available and that, in the interim, people are asked “to refrain from speculation regarding the cause” of the outbreak. News reports suggested that some of the food served during the reunion weekend may have been taken to people’s homes as leftovers. Meanwhile, a variety of food samples from the reunion are being tested and state and local public health officials are trying to track down where the E. coli bacteria may have come from. “While we are narrowing down the source, it is too early to tell what the exact cause of the outbreak was,” noted the county’s Facebook post. “Local food vendors have been very cooperative in this investigation. We encourage anyone who attended the event to not consume any leftover foods they may have brought back from the event and to contact their local health department if they still have leftover food, in case it may become of interest.” The Dawson County Health Department and the Montana Department of Public Health and Human Services are working together on the outbreak. Infection with E. coli bacteria often causes several stomach cramps and diarrhea (often bloody) and vomiting. Fever may be present, but in some cases, it is not. Symptoms usually appear within two to eight days after exposure. Most people get better within five to seven days. Some infections are very mild, but others are severe or even life-threatening, particularly if the person develops the kidney-related complication known as HUS (hemolytic uremic syndrome).
]]> “We have approximately 25 cases under investigation. It is possible the investigation may determine some of the 25 cases are unrelated, and it is also possible more cases may be reported to us in the coming days,” said Polly Carver-Kimm, communications director for the Iowa Department of Public Health (IDPH). The store-made “Zesty Potato Salad” and “Traditional Potato Salad” dated July 14-24 were removed from sale and Big G is cooperating with the outbreak investigation. According to the Iowa Department of Inspections and Appeals (DIA), neither of the potato salads has been sold to the public since July 22. “They’re doing testing, and some sort of cross-contamination is something they’re thinking is possible,” Gary Grafft, co-owner of the store, told Food Safety News on Thursday. He said the family-owned, single-location store has not had a similar incident since the deli opened in 1978. He told the Iowa City Press-Citizen newspaper that a firm from Cedar Rapids, IA, was coming in Thursday to do a deep cleaning and sanitizing of the deli. Two Iowa state agencies issued a joint consumer advisory Monday warning the public about the Big G deli’s potato salads and citing the risk of Salmonella infection. “The bottom line is that no one should eat this product,” IDPH Medical Director Dr. Patricia Quinlisk said in the public warning. “If you have it in your refrigerator, you should throw it away.” Big G reportedly posted the following statement on its Facebook page on Tuesday, however, it was no longer there on Thursday:
“We have approximately 25 cases under investigation. It is possible the investigation may determine some of the 25 cases are unrelated, and it is also possible more cases may be reported to us in the coming days,” said Polly Carver-Kimm, communications director for the Iowa Department of Public Health (IDPH). The store-made “Zesty Potato Salad” and “Traditional Potato Salad” dated July 14-24 were removed from sale and Big G is cooperating with the outbreak investigation. According to the Iowa Department of Inspections and Appeals (DIA), neither of the potato salads has been sold to the public since July 22. “They’re doing testing, and some sort of cross-contamination is something they’re thinking is possible,” Gary Grafft, co-owner of the store, told Food Safety News on Thursday. He said the family-owned, single-location store has not had a similar incident since the deli opened in 1978. He told the Iowa City Press-Citizen newspaper that a firm from Cedar Rapids, IA, was coming in Thursday to do a deep cleaning and sanitizing of the deli. Two Iowa state agencies issued a joint consumer advisory Monday warning the public about the Big G deli’s potato salads and citing the risk of Salmonella infection. “The bottom line is that no one should eat this product,” IDPH Medical Director Dr. Patricia Quinlisk said in the public warning. “If you have it in your refrigerator, you should throw it away.” Big G reportedly posted the following statement on its Facebook page on Tuesday, however, it was no longer there on Thursday: 
 Salmonella infection is relatively common in the United States and has been linked to eating eggs, poultry, meat, unpasteurized milk or juice, cheese, raw fruits and vegetables, spices and nuts. According to CDC, most people infected with the bacteria develop diarrhea, fever, and abdominal cramps between 12 and 72 hours after infection. The illness usually lasts four to seven days, and most individuals recover without treatment. However, CDC states that in some cases diarrhea may be so severe that the patients need to be hospitalized. In these patients, the Salmonella infection may spread from the intestines to the bloodstream, and then to other parts of the body. In these cases, Salmonella can cause death unless the person is treated promptly with antibiotics. The elderly, infants, and those with impaired immune systems are more likely to have a severe illness.
Salmonella infection is relatively common in the United States and has been linked to eating eggs, poultry, meat, unpasteurized milk or juice, cheese, raw fruits and vegetables, spices and nuts. According to CDC, most people infected with the bacteria develop diarrhea, fever, and abdominal cramps between 12 and 72 hours after infection. The illness usually lasts four to seven days, and most individuals recover without treatment. However, CDC states that in some cases diarrhea may be so severe that the patients need to be hospitalized. In these patients, the Salmonella infection may spread from the intestines to the bloodstream, and then to other parts of the body. In these cases, Salmonella can cause death unless the person is treated promptly with antibiotics. The elderly, infants, and those with impaired immune systems are more likely to have a severe illness.
(To sign up for a free subscription to Food Safety News, click here.)
]]>